All Obese Patients Should Be Screened For ADHD
 Regular readers will recall previous posts on the association between attention deficit disorder (ADD) and obesity.
Regular readers will recall previous posts on the association between attention deficit disorder (ADD) and obesity.
As this condition significantly affects impulse control, ability to plan, perseverance, time management, and many other factors and skills essential for weight management, this relationship should be no surprise.
In our own clinical experience (as suggested in several recent publications from others), managing ADD can often be the key step to managing weight gain.
Once you start systematically screening patients for ADD in an obesity clinic, it seems to be present in a surprisingly large number – almost 20-30%.
This number is consistent with the findings of another study, this time by Bruno Palazzo Nazar and colleagues from the Federal University of Rio de Janeiro, Brazil, published in the Journal of Attention Disorders.
The study sample consisted of women seeking nonsurgical treatment of obesity at a public endocrinology hospital with an eating disorders and obesity clinic, in Rio de Janeiro.
One hundred and fify-five consecutive patients presenting in the clinic were approached for this study. Exclusion criteria included less than 5 years of schooling/inability to read and fill out forms and questionnaires; current alcohol or drug abuse, history of bipolar or psychotic disorder; current treatment with psychoactive drugs; and presence of uncontrolled clinical, neurological, or endocrine disorders, especially if they interfere with weight, appetite, and attention; and patients older than 60 years.
Based on a battery of validated questionnaires and semi-structured interviews, 28.3% of patients were diagnosed with ADD, which, in turn, was significantly correlated with more severe binge eating, bulimic behaviors, and depressive symptoms.
As the authors note, this rate of almost 30% is far higher than the expected rate of less than 5% in the general population.
In fact, given the rather rigorous exclusion criteria, the actual prevalence of ADHD in this patient set may actually be even higher.
As a clinician, I’d certainly support the notion that we should be aware of the high prevalence of ADHD in patients presenting in obesity programs. Making this diagnosis and managing this issue, may make all the difference in long-term outcomes.
AMS
Cambridge, UK
photo credit: Peter Vidrine via photo pin cc
![]() Nazar BP, Pinna CM, Suwwan R, Duchesne M, Freitas SR, Sergeant J, & Mattos P (2012). ADHD Rate in Obese Women With Binge Eating and Bulimic Behaviors From a Weight-Loss Clinic. Journal of attention disorders PMID: 22930790
Nazar BP, Pinna CM, Suwwan R, Duchesne M, Freitas SR, Sergeant J, & Mattos P (2012). ADHD Rate in Obese Women With Binge Eating and Bulimic Behaviors From a Weight-Loss Clinic. Journal of attention disorders PMID: 22930790
.
Training Obesity Specialists from Around the Globe
 As regular readers may recall, I am one of the founding faculty of the International Association for the Study of Obesity’s (IASO) obesity specialist certification program (SCOPE), where I will once again be presenting several sessions on obesity assessment and management to an audience of physicians and allied health professionals from around the world (I wonder how many countries will be represented this time around).
As regular readers may recall, I am one of the founding faculty of the International Association for the Study of Obesity’s (IASO) obesity specialist certification program (SCOPE), where I will once again be presenting several sessions on obesity assessment and management to an audience of physicians and allied health professionals from around the world (I wonder how many countries will be represented this time around).
As members of the Canadian Obesity Network are probably aware, membership in CON now includes a number of important benefits, one of them being reduced registration costs for IASO events – like SCOPE.
I know for sure that at least one Canadian will be at this course (Christian, who also happens to be my post-doc fellow), but there well may be others.
I certainly look forward to another intense course of teaching and learning.
AMS
Cambridge, UK
What Are Patients Eating One Year After Gastric Banding?
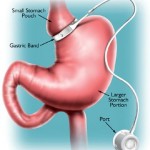 There is no doubt that, for severe obesity, bariatric surgery is currently the most effective treatment with well-documented benefits on a wide range of health problems and quality of life.
There is no doubt that, for severe obesity, bariatric surgery is currently the most effective treatment with well-documented benefits on a wide range of health problems and quality of life.
However, as regular readers will recall, I am also the first to point out that surgery is not a ‘stand-alone’ procedure, but rather, requires intense and probably life-long follow-up to prevent nutritional and psychological complications that may emerge even years after surgery.
This problem is again emphasized by a recent study by McGrice and Porter, from Melbourne, Australia, published in OBESITY SURGERY.
They invited 215 patients to complete validated Food Frequency Questionnaire 12 months following adjustable gastric banding.
Although only bout 25% of patients responded, those who did, reported daily energy intakes ranging between just 270 (???) to over 3000 Calories, with an average of about 1200 Cal/day.
Not surprisingly, many patients failed to meet even the minimum recommendations for macronutrient intake.
The average fibre intake was only 14 g/day (current recommendations are 25 g for women and 30 g for men) and the average diet contained 36% total fat (almost half of which was saturated fat) compared to the recommendations of 20-25%.
From these findings the authors conclude that:
“Patients’ dietary intakes vary significantly 1 year post-laparoscopic adjustable band surgery, with many patients not meeting recommendations. These results suggest that patients decrease their total energy intake; however, eating habits may not improve as they consume a diet high in saturated fat. It is recommended that all patients receive dietary education about diet quality post-laparoscopic adjustable gastric band surgery to assist them in improving their diet quality as well as quantity for optimal health and weight loss.”
Critics will immediately point out the important limitations of this study including the rather low response rate and the rather ‘iffy’ reliability of one-time questionnaires.
But, unless we assume that only people with post-surgical nutritional issues responded to this questionnaire, we must assume that there are severe nutritional issues in a considerable number of patients one year after surgery.
Although this study is limited to patients undergoing gastric banding, we have yet to see data on long-term nutrition intake in patients undergoing other types of bariatric surgery.
Certainly this study (despite its limitations), if nothing else, should serve as a reminder that we need to pay long-term attention to dietary intake with periodic and ongoing assessment of nutritional status in patients undergoing bariatric surgery.
In fact, I would not be surprised if those who lose the most weight (and by surgical standards, would be considered to have the greatest ‘success’), are the ones at highest risk for nutritional problems.
If you have experienced nutritional problems or deficiencies post-surgery, I’d certainly love to hear your story.
AMS
Edmonton, Alberta
![]() McGrice MA, & Porter JA (2012). What are Gastric Banding Patients Eating One Year Post-Surgery? Obesity surgery PMID: 22923340
McGrice MA, & Porter JA (2012). What are Gastric Banding Patients Eating One Year Post-Surgery? Obesity surgery PMID: 22923340
.
4-6 Months of Exclusive Breast Feeding Associated With Lower Obesity Risk Across 8 European Countries
 Regular readers may recall a recent post on the efforts to increase breast feeding in Canada. Notably, a recent media report had NYCs Mayor Bloomberg (of banning oversize pop fame) to demand that clinics lock up baby formula in an attempt to increase breast feeding, as a further measure to prevent obesity.
Regular readers may recall a recent post on the efforts to increase breast feeding in Canada. Notably, a recent media report had NYCs Mayor Bloomberg (of banning oversize pop fame) to demand that clinics lock up baby formula in an attempt to increase breast feeding, as a further measure to prevent obesity.
But how long should infants be breast fed to prevent obesity?
This question was addressed in a paper by Hunsberger and colleagues from 8 European countries (Italy, Estonia, Cyprus, Belgium, Sweden, Hungary, Germany and Spain) published in Public Health Nutrition.
The researchers assessed the potential impact of exclusive breast-feeding on childhood overweight in cross-sectional datathe longitudinal cohort study IDEFICS. The analysis included 14 726 children aged 2-9 years for whom early feeding practices were reported by parents in standardized questionnaires.
After controlling for education, income and other potential confounders, breast-feeding exclusively for 4-6 months were about 30% less likely to be overweight when compared with children never exclusively breast-fed. This association could not be explained by socio-economic characteristics or maternal overweight.
Thus, the authors conclude that exclusive breast-feeding for 4-6 months may confer protection against overweight in addition to other known benefits. Exclusive breast feeding beyond 6 months did not appear to have any additional benefit in this regard.
While association studies do not prove causality, there are certainly a number of biologically plausible hypotheses that would explain the beneficial effect of breast feeding on the development of childhood obesity, so certainly this is a finding that should not be simply dismissed.
I wonder though how feasible exclusive breastfeeding for 4-6 months would actually be in most settings.
AMS
Edmonton, Alberta
photo credit: c r z via photo pin cc
![]() Hunsberger M, Lanfer A, Reeske A, Veidebaum T, Russo P, Hadjigeorgiou C, Moreno LA, Molnar D, De Henauw S, Lissner L, & Eiben G (2012). Infant feeding practices and prevalence of obesity in eight European countries – the IDEFICS study. Public health nutrition, 1-9 PMID: 22916704
Hunsberger M, Lanfer A, Reeske A, Veidebaum T, Russo P, Hadjigeorgiou C, Moreno LA, Molnar D, De Henauw S, Lissner L, & Eiben G (2012). Infant feeding practices and prevalence of obesity in eight European countries – the IDEFICS study. Public health nutrition, 1-9 PMID: 22916704
.
Pulse Grain Consumption Lowers Obesity Risk?
 Generally speaking, I am wary of ‘magical’ properties of foods.
Generally speaking, I am wary of ‘magical’ properties of foods.
Humans have certainly evolved to survive on a wide range of foods and diets, which is why, as a species, we have managed to live in virtually every climate and geographic corner of this planet.
Nevertheless, nutrition science has taught us much about how exactly certain foods or nutrients affect metabolism in ways that other foods may not.
One food that may not have yet received the attention it deserves in this regard are pulses, incidently a staple of many traditional diets – for e.g. in India, no meal is complete without a ‘dal’ dish – in fact, ‘dal-roti’ is a widely accepted colloquial term for ‘food’.
It also turns out that pulses are one of Canada’s major ‘cash crops’ (who would have guessed?).
For readers wanting to learn more about the potential health benefits of eating pulses, I wish to point you to a recent review article by Christopher Marinangeli and Peter Jones from the Richardson Centre for Functional Foods and Nutraceuticals, Winnipeg, Manitoba, published in the British Journal of Nutrition.
The paper deals in depth with the effect of pulse grains on energy expenditure, substrate oxidation, body composition, fat deposition and satiety.
These effects are mediated by a wide range of known components of pulse grains, including pulse-derived fibre and resistant starch, have been shown to alter energy expenditure, substrate trafficking and fat oxidation as well as visceral adipose deposition. The high content of amino acids like arginine and glutamine may produce thermogenic effects. Finally, evidence suggests that pulse-derived fibres, trypsin inhibitors and lectins may reduce food intake by inducing satiety via facilitating and prolonging cholecystokinin secretion.
However, the authors also point out that many of these effects have so far only been demonstrated in rather small experimental (efficacy) studies with notably few high-quality mechanistic studies, not to mention the lack of clinical ‘feeding’ studies.
Nevertheless, pulse grains may represent an understudied nutrient and energy source that could potentially assist both in the prevention and management of obesity.
Even before such data is available I would not be surprised to see pulses declared as the next ‘super foods’ and supplement stores peddling ‘pulse extracts’ for their putative (and probably widely overblown) health benefits.
On a personal note however, I am all for pulse grains and can personally attest to the satiety enhancing impact of adding pulses to my diet.
AMS
Vancouver, BC
photo credit: Dey via photo pin cc
Marinangeli CP, & Jones PJ (2012). Pulse grain consumption and obesity: effects on energy expenditure, substrate oxidation, body composition, fat deposition and satiety. The British journal of nutrition, 108 (S1) PMID: 22916815
.


