Why Obesity Is A Chronic Disease
 While I am currently teaching at a Harvard Medical School course on obesity for obesity educators here in Las Vegas, I thought it may be appropriate to post a link to my recent Obesity Canada webinar (about 60 mins) on why obesity is a chronic disease.
While I am currently teaching at a Harvard Medical School course on obesity for obesity educators here in Las Vegas, I thought it may be appropriate to post a link to my recent Obesity Canada webinar (about 60 mins) on why obesity is a chronic disease.
The full video can be accessed by clicking here
@DrSharma
Las Vegas, NV
Time To Change The Obesity Narrative
 This week, I once again presented on the need for recognising obesity as a chronic disease at the annual European Society for the Study of Obesity Collaborating Centres for Obesity (EASO-COMs) in Leipzig, Germany.
This week, I once again presented on the need for recognising obesity as a chronic disease at the annual European Society for the Study of Obesity Collaborating Centres for Obesity (EASO-COMs) in Leipzig, Germany.
Coincidently, The Lancet this week also published a commentary (of which I am a co-author) on the urgent need to change the obesity “narrative”.
So far, the prevailing obesity “narrative” is that this is a condition largely caused by people’s lifestyle “choices” primarily pertaining to eating too much and not moving enough, and that this condition can therefore be prevented and reversed simply by getting people to make better choices, or in other words, eating less and moving more.
As pointed out in the commentary, this “narrative” flies in the face of the overwhelming evidence that obesity is a rather complex multi-factorial heterogenous disorder, where long-term success of individual or population-based “lifestyle” interventions can be characterised as rather modest (and that is being rather generous).
This is not to say that public health measures targeting food intake and activity are not important – but these measures go well beyond “personal responsibility”
” The established narrative on obesity relies on a simplistic causal model with language that generally places blame on individuals who bear sole responsibility for their obesity. This approach disregards the complex interplay between factors not within individuals’ control (eg, epigenetic, biological, psychosocial) and powerful wider environmental factors and activity by industry (eg, food availability and price, the built environment, manufacturers’ marketing, policies, culture) that underpin obesity. A siloed focus on individual responsibility leads to a failure to address these wider factors for which government policy can and should take a leading role. Potential health-systems solutions are also held back by insufficient understanding of obesity as a chronic disease and of the necessary integration across specialties.“
It is also important to recognise that the prevailing “lifestyle” narrative plays a major role in the issue of weight-bias and discrimination:
“Behind every obesity statistic are real people living with obesity. The prevailing narrative wrongly portrays people with obesity in negative terms as “guilty” of obesity through “weakness” and “lack of willpower”, succumbing to the siren call of fast and other poor food choices. This narrative leads to stigmatisation, discrimination—including in health services, employment, and education—and undermines individual agency.“
Thus, it is time to change this narrative:
“If the narrative is instead reframed around individuals at risk of or living with obesity as protagonists with agency who operate within physiological limitations and a much larger obesogenic environment over which their control is limited, then a better, more accurate story can be told.“
This new narrative must incorporate four dimensions.
“First, recognise that obesity requires multiple discrete actors and sectors to work together simultaneously through many entry points. Second, change the words and images used to portray obesity to shift blame away from individuals and towards upstream drivers. For example, photographs of anonymous or faceless people with obesity must be substituted with images of real people that foster respect and identification. Third, prioritise childhood obesity and the growing burden of obesity in low-income settings. Rights-based policy approaches that address inequalities and social and physical determinants of obesity are particularly relevant. Finally, appreciate that obesity is a chronic disease within the health system, with both its prevention and management embedded within calls for effective and comprehensive universal health coverage globally.“
Following this line of reasoning we argue that,
“Shifting to a human-focused narrative that encompasses this vulnerability and complexity will require effort and commitment across many sectors. We call on all affected by or concerned with obesity to come together with a common sense of purpose and shared accountability for building this new narrative and a more comprehensive response to obesity.“
Not discussed in this paper (largely due to space limitation), is my pet peeve, that we also need a new non-anthropometric definition of obesity – one that relies on actual health measures rather than just scales and measuring tapes. As we move to a “disease” definition of obesity, we need to ensure that we are not mis-labeling healthy individuals as “diseased” just because they happen to exceed a certain body weight, as well as the corollary, mis-labeling individuals who may stand to benefit from obesity treatments as not having obesity just because they fall below an arbitrary BMI cutoff.
@DrSharma
Leipzig, Germany
The Clinical Importance of Using People-First Language in Obesity Management
 Regular readers should by now be well aware of the importance of using people-first language when referring to people living with obesity (as in “patient with obesity” not “obese patient”).
Regular readers should by now be well aware of the importance of using people-first language when referring to people living with obesity (as in “patient with obesity” not “obese patient”).
As I have noted in previous posts, living with a condition is not the same as being defined by that condition – this is why we do not refer to people living with dementia or cancer as being “demented” or “cancerous”.
As elegantly pointed out by Lee Kaplan in a presentation he delivered at the Harvard Medical School Center For Global Health Delivery consultation on obesity, currently being held in Dubai, there is also a pressing clinical argument for speaking of obesity as a disease rather than a descriptor of a “state”.
Take for e.g. the case of a patient with hypertension, who, thanks to effective on-g0ing anti-hypertensive treatment has managed to control his blood pressure levels over the past 10 years. In fact, at no time in the past 10 years has the patient ever presented with elevated blood pressure. Any clinician would agree that this patient would still be declared to have “hypertension” despite currently not being “hypertensive”.
Similarly, a patient whose depression is well-controlled with an anti-depressant is still a patient living with “depression”, although they are not currently “depressed”.
Likewise, we can probably all agree that a patient who has undergone coronary bypass surgery, is still someone living with coronary artery disease, even if they have not experienced a single angina pectoris attack since their surgery.
In all of these cases, hypertension, depression, and coronary artery disease would continue to appear on their medical problem list.
When applied to obesity, this means that even if someone has successfully managed to lose their weight to a level that they are no longer clinically “obese”, they are still someone living with “obesity”. Even if their BMI (not a good measure of obesity) should drop to below 25, they are still someone living with obesity (albeit, as in the case of the above examples, living with “controlled” or “treated” obesity).
Thus, they continue to have “obesity” even if they are currently not “obese” – ergo, the diagnosis “obesity” should remain on their problem list.
Furthermore, given the high rates of recidivism, keeping obesity on the problem list serves as an important reminder to the clinician to continue supporting and reinforcing ongoing obesity treatment (even if this treatment is only behavioural).
It should be evident from this analogy that although the use of people-first language may seem like semantics, it does have very real consequences for long-term clinical management.
@DrSharma
Dubai, UAE
The Heterogeneity of Obesity
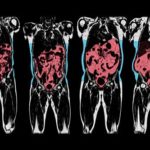 In the same manner in that there is not one predisposing factor for the development of obesity, the phenotypic clinical presentation of obesity is likewise extraordinarily heterogenous. (This has some authors speaking of “obesities” rather than “obesity”).
In the same manner in that there is not one predisposing factor for the development of obesity, the phenotypic clinical presentation of obesity is likewise extraordinarily heterogenous. (This has some authors speaking of “obesities” rather than “obesity”).
While it is now well established that BMI is a measure of size rather than health, it is perhaps less well recognised how the different types of body fat and their storage in various fat depots and organs can contribute to cardiometabolic disease (location, location, location!).
Now, a comprehensive review by Ian Neeland from the University of Texas Southwestern Medical Center, Dallas, together with my colleagues Paul Poirier and JP Despres from Laval University in Quebec, published in Circulation discusses the cardiovascular and metabolic heterogeneity of obesity.
As the authors point out,
“Although the BMI has been a convenient and simple index to monitor the growth in obesity prevalence at the population level, many metabolic and clinical studies have revealed that obesity, when defined on the basis of the BMI alone, is a remarkably heterogeneous condition. For instance, patients with similar body weight or BMI values have been shown to display markedly different comorbidities and levels of health risk.”
Not only has BMI never emerged as a significant component in risk engines such as the Framingham risk score, there are many individuals with obesity who never develop metabolic complications or heart disease during the course of their life.
The paper offers a good review of what the author describe as adipose dysfunction or “adiposopathy” = “sick fat”. Thus, in some individuals, there is an accumulation of “unhealthy” fat (particularly visceral and ectopic fat), whereas in others, excess fat predominantly consists of “healthy” fat (predominantly in subcutaneous depots such as the hips and thighs).
The authors thus emphasise the importance of measuring fat location with methods ranging from simple anthropometric measures (e.g. waist circumference) to comprehensive imaging techniques (e.g. MRI).
The authors also provide a succinct overview of exactly how this “sick fat” contributes to cardiometabolic risk and briefly touches on the behavioural, medical, and surgical management of patients with obesity and elevated cardiometabolic risk.
I, for one, was also happy to see the inclusion of the Edmonton Obesity Staging System in their reflections on this complex issue.
This paper is certainly suggested reading for anyone interested in the link between obesity and cardiovascular disease.
@DrSharma
Edmonton, AB
Free Webinar: Assessing Body Fat – Taking Obesity Phenotyping To The Next Level
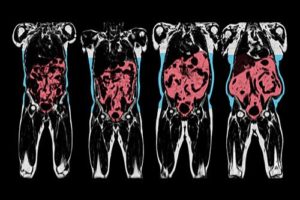 As readers will be well aware, n terms of health risks, fat is not fat is not fat is not fat.
As readers will be well aware, n terms of health risks, fat is not fat is not fat is not fat.
Rather, whether or not body fat affects health depends very much on the type of body fat and its location.
While there have been ample attempts at trying to describe body fat distribution with simple anthropometric tools like measuring tapes and callipers, these rather crude and antiquated approaches have never established themselves in clinical practice simply because they are cumbersome, inaccurate, and fail to reliably capture the exact anatomical location of body fat. Furthermore, they provide no insights into ectopic fat deposition – i.e. the amount of fat in organs like liver or muscle, a key determinant of metabolic disease.
Recent advances in imaging technology together with sophisticated image recognition now offers a much more compelling insight into fat phenotype.
In this regard, readers may be interested in a live webinar that will be hosted by the Canadian Obesity Network at 12.00 pm Eastern Standard Time on Thu, Nov 23, 2017. The webinar provides an overview of a new technology developed by the Swedish company AMRA, that may have both important research and clinical applications.
The talk features Olof Dahlqvist Leinhard, PhD, Chief Scientific Officer & Co-Founder at AMRA and Ian Neeland, MD, a general cardiologist with special expertise in obesity and cardiovascular disease, as well as noninvasive imaging at the UT Southwestern Medical Center in Dallas, US.
Registration for this seminar is free but seats are limited.
To join the live event register here.
I have recently heard this talk and can only recommend it to anyone interested in obesity research or management.
@DrSharma
Edmonton, AB


