The Effects of Obesity on Skeletal Muscle Contractile Function

Skeletal muscle
Given that obesity has profound effects on all organ systems, it is not surprising that excess body fat is also associated with a decrease in muscle function.
The complex biology of the molecular, structural, and functional changes that have been associated with obesity are now extensively discussed in a review article by James Tallin and colleagues, published in the Journal of Experimental Biology.
Without going into the molecular details here, suffice it to say that there is considerable evidence to show and explain why muscular function is impaired in both animal models and humans with excess body fat. (For e.g. at a cellular level, the dominant effects of obesity are disrupted calcium signalling and 5′-adenosine monophosphate-activated protein kinase (AMPK) activity. As a result, there is a shift from slow to fast muscle fibre types. There is also evidence for an impairment in myogenesis resulting from disruption of muscle satellite cell activation. Furthermore, muscle function is affected by insulin resistance and decreased adiponectin levels generally associated with obesity).
Although individuals with obesity will often have a larger muscle mass and may well be stronger than “normal-weight” individuals, when corrected for the amount of extra muscle, it is evident that the muscles are less efficient.
In fact, many of the biochemical and structural changes that occur in obesity are very similar to those found with aging. Not surprisingly, when aging meets obesity, things get even worse.
Although the paper does not discuss the reversibility of these changes with weight loss (or obesity treatment in general), I am aware of other data showing that much of the loss of muscle contractile function associated with obesity can be reversed with weight loss.
A clinical correlate of this is the fact that, following weight loss, individuals often find that it takes far more exercise to burn the same number of calories than before (this is not just because the person is now carrying less weight).
Given the increased recognition that lean body mass is an important determinant of overall health and function, clearly this topic is of continuing interest.
@DrSharma
Edmonton, AB
Applying The Etiological Framework For Obesity Assessment In Clinical Practice
 Continuing with citations from my article in Obesity Reviews on an aeteological framework for assessing obesity, that guides us through a systematic assessment of factors influencing energy metabolism, ingestive behaviour, and physical activity, we ca now apply this framework in clinical practice:
Continuing with citations from my article in Obesity Reviews on an aeteological framework for assessing obesity, that guides us through a systematic assessment of factors influencing energy metabolism, ingestive behaviour, and physical activity, we ca now apply this framework in clinical practice:
This paper provides a comprehensive framework, which should enable clinicians to systematically assess and identify the socio‐cultural, biophysical, psychological and iatrogenic determinants of increased energy intake and reduced energy expenditure in patients presenting with excess weight or weight gain. Beginning with an assessment of energy requirements and metabolism, clinicians should systematically assess the role and determinants of ingestive and activity behaviour to identify the factors promoting positive energy balance. This will enable clinicians to develop management plans that address the root causes of weight gain and move beyond the simplistic and generally ineffective recommendation to ‘eat less and move more’.
Thus for example, in a listless patient ‘self‐medicating’ with food, identification and treatment of depression may be the first step to reducing food intake and preventing further weight gain. In a patient with socioeconomic barriers to healthy eating or physical activity, referral to a social worker who can assist in identifying and accessing community resources may be important. Identification and effective treatment of obstructive sleep apnoea may be the key to increasing activity in someone with this disorder. Psychological counselling to manage alcohol or substance abuse or to help patients deal with binge eating resulting from past trauma, emotional neglect or grief, can put patients on a path to successful weight management. Clearly, the common notion that all forms of obesity can be addressed simply by counselling patients on diet and exercise should be considered ineffective and obsolete.
To conclude this series, we will tomorrow look at some of the potential limitations of this system.
@DrSharma
Edmonton, AB
Factors Affecting Energy Metabolism: Brown Fat, Medications, Weight Loss
 Continuing with citations from my article in Obesity Reviews on an aeteological framework for assessing obesity, we now turn to the some of the factors that can affect metabolic rate:
Continuing with citations from my article in Obesity Reviews on an aeteological framework for assessing obesity, we now turn to the some of the factors that can affect metabolic rate:
Metabolically Active Fat
Recent evidence suggests that brown adipose tissue (BAT) exists into adult hood and can, when present account for as much as 20% of daily resting energy expenditure. While the exact contribution of BAT (or lack thereof) to obesity remains to be determined, the presence and inducibility of BAT by cold exposure is inversely related to BMI, appears higher in women, and diminishes with aging. Given the role of cold exposure in the expression of BAT, it can be speculated that an increase in ambient temperature may promote weight gain by significantly reducing BAT and, thus, metabolic rate in some individuals. In rodents, increased production of neuropeptide Y in the hypothalamus can not only increase food intake but also reduce energy expenditure via a reduction in non‐shivering thermogenesis in BAT and facilitate triglyceride deposition through increased insulin levels.
Medications
A wide range of medications can affect metabolic rate. Notably, the use of beta‐blockers has been shown to significantly reduce thermogenesis, resulting in clinically relevant weight gain 34. Metabolic rates can also be reduced by the discontinuation of drugs that promote thermogenesis such as beta‐adrenergic agents, stimulants (including performance‐enhancing and illicit drugs like crack/cocaine), coffee or nicotine, resulting in weight gain.\
Weight Loss
Finally, weight loss can markedly reduce energy requirements with a 5–10% reduction in body weight reducing resting metabolic rate by as much as 20% in some individuals, thereby substantially increasing the susceptibility to weight regain in the post‐obese state.
Commentary: In summary, any of the many factors that can reduce metabolic rate, can result in weight gain even with no change in energy intake or energy expenditure. In a clinical setting, this would apply to the patient, who tells you that they have not changed their food intake or their activity levels and, yet, have gained weight. Rather than simply discarding this information from a patient as being untrue or “delusional”, clinicians should give careful consideration to the factor that there very well may be factors that have led to a significant reduction in metabolic requirements.
@DrSharma
London, ON
Factors Affecting Energy Metabolism: Sarcopenia
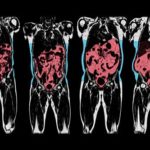 Continuing with citations from my article in Obesity Reviews on an aeteological framework for assessing obesity, we continue discussing factors that can affect metabolic rate:
Continuing with citations from my article in Obesity Reviews on an aeteological framework for assessing obesity, we continue discussing factors that can affect metabolic rate:
Sarcopenia
The importance of fat-free mass as the key determinant of resting metabolic rate, even in a very obese individual [sic], cannot be over emphasized. Obese individuals [sic] can present with wide variations in lean body mass, almost entirely accounted for by differences in skeletal muscle mass. Thus, any change in muscle mass can markedly affect basal energy requirements. In this context it is important to remember that in ambulatory individuals, the mass of weight‐bearing muscles is directly proportional to BMI, as heavier individuals require a greater skeletal muscle mass to support and move their excess weight. This alone accounts for much of the higher basal and activity‐related energy requirements of larger individuals.
Although inactivity may be the most common cause of decreased skeletal muscle mass and reduced basal metabolic needs in obese individuals [sic], it is important to consider other causes of muscular atrophy that can likewise markedly reduce energy demands. A wide range of nutritional, neuromuscular, endocrine, renal, cardiac, pulmonary, inflammatory, infectious or neoplastic conditions can result in muscular wasting and sarcopenia. Reduced skeletal muscle mass and weight gain is also noted after many cancer treatments, although the mechanisms remain unclear. Any reduction in skeletal muscle mass not accounted for by a decrease in physical activity and ambulation should prompt investigations for other causes of muscular wasting.
Commentary: Sarcopenic obesity is perhaps even more prevalent than most people may think – especially in people who have slight overweight or even moderate obesity. It is particularly common in certain ethnic groups such as South Asians, even at “normal” BMIs. Clinically, this is where body composition studies can be helpful. Although a reduction in muscle mass does reduce resting metabolic rate (RMR), it is important to remember that overall skeletal muscle only accounts for about 15% of RMR. This is why, the notion that building up muscle mass will help with weight loss by burning more calories is not really an effective weight loss strategy.
Factors Affecting Energy Metabolism: Neuroendocrine Factors
 Continuing with citations from my article in Obesity Reviews on an aeteological framework for assessing obesity, we continue discussing factors that can affect metabolic rate:
Continuing with citations from my article in Obesity Reviews on an aeteological framework for assessing obesity, we continue discussing factors that can affect metabolic rate:
Neuroendocrine Factors
Across the entire age continuum, a wide range of neuroendocrine factors can not only affect metabolic rate, but also substrate partitioning and utilization, which may directly or indirectly contribute to weight gain. The latter point is of particular significance as low rates of fat oxidation are associated with an increased risk of weight gain.
A wide range of neuroendocrine hormones and biomarkers can affect energy metabolism; sympathetic nervous system activity and thyroid function are two major factors directly influencing resting energy expenditure.
Sympathetic nervous activity is also a major determinant of post‐prandial thermogenesis and the thermogenic response to a glucose load has been shown to be significantly lower in obese [sic] individuals, a finding that persists even with substantial weight loss.
Specific examples of endocrine hormones that affect energy metabolism and substrate partitioning include cortisol, growth hormone (GH) and testosterone.
Catabolism associated with hypercortisolism or Cushing’s syndrome can reduce energy requirements and increase the deposition of truncal fat.
Discontinuation of GH treatment at the end of childhood growth in individuals with GH deficiency markedly increases fat mass and decreases metabolic rate, whereas GH treatment in GH‐deficient adults has beneficial effects on protein metabolism, energy expenditure and thyroid metabolism.
Testosterone deficiency can also result in abnormal energy partitioning, which adversely alters anabolism and reduces metabolic rate.
It is important to note that a careful history and physical examination should precede any endocrine testing for these disorders, as testing should be reserved for patients with an above‐normal pretest probability for one of these conditions.
Commentary: as pointed out in the last paragraph, while all of the above are important considerations, each one is quite rare, which is why it is important to use clinical judgement in recognising these factors, rather than simply ordering a battery of endocrine tests on every patient.
@DrSharma
London, ON


