Why Weight-Loss Challenges Send the Wrong Message
 I was recently, once again asked about my opinion on weight-loss challenges. So here is a repost of an article I wrote back in 2008 on this topic – apparently, it is still as relevant today, as it was almost a decade ago.
I was recently, once again asked about my opinion on weight-loss challenges. So here is a repost of an article I wrote back in 2008 on this topic – apparently, it is still as relevant today, as it was almost a decade ago.
There appears to be a rather widespread notion out there that introducing a bit of competition into the affair may spurn people on to try and lose those “extra” pounds.
In fact, a quick google search on the term “weight-loss challenge” reveals an amazing array of challenges from voyeuristic and sadistic TV shows like the “Biggest Loser” to well-meant workplace wellness initiatives or fund raisers. I am sorry to admit that I recently even became aware of a weight-loss challenge within my own hospital – well intended, but useless in the fight against obesity.
So what’s wrong with this idea? Isn’t competition a great motivator?
Sure it is – and people will do anything to win a competition – including crazy stuff like starve themselves, exercise till they drop, or even (God forbid) pop diet pills, diuretics or laxatives just to win.
All of this is in direct contradiction to a fundamental principle of obesity management: you do not do things to lose weight that you are unlikely to continue doing to keep the weight off.
Most people seem to think that if only they could lose some weight, they will somehow be able maintain that lower body weight in the long-term with less effort.
The reality unfortunately is (and most dieters have experienced this over and over again) that no matter what diet or exercise routine you chose, no matter how slow or fast you lose the weight, no matter how long you keep the weight off – the minute you relax your efforts, the weight simply comes back.
As I have blogged before: obesity is a chronic disease for which we have no cure – only treatments! When you stop the treatment the weight (and any related problem) simply comes back.
By now you will already have figured out the problem with these challenges – unless you are very modest and reasonable about your weight-loss target and are carefully making changes that you can reasonably sustain forever, you are simply setting yourself up for failure.
If you are indeed modest and reasonable – you’ve already lost the competition to all the crazy folks who’ll do anything just to win.
My advise to anyone with a weight problem – the next time you see an invitation to a weight-loss challenge – simply ignore it!
If you really think you will benefit from obesity treatment – seek help from a trained and accredited health professional with experience in weight management – let’s put an end to weight cycling!
@DrSharma
Berlin, Germany
Conflict Disclosures In Nutrition Research
 As someone who has often engaged in research projects, consultation, or speaking engagements sponsored or otherwise supported by industry (all of which I happily acknowledge and declare), I am a keen observer of the ongoing discussion about when and how researchers need to be wary of potential biases and conflicts.
As someone who has often engaged in research projects, consultation, or speaking engagements sponsored or otherwise supported by industry (all of which I happily acknowledge and declare), I am a keen observer of the ongoing discussion about when and how researchers need to be wary of potential biases and conflicts.
As I pointed out in previous posts, among all of the potential conflicts, the financial one is perhaps the easiest to declare and otherwise manage.
A recent article by John Ionnadis and John Trepanowski, published in JAMA, discusses the wide range of conflicts (most of which may be non-financial), that one may wish to have declared and exposed, especially when it comes to nutrition research.
The authors single our nutrition research for good reasons:
“…the totality of an individual’s diet has important effects on health, most nutrients and foods individually have ambiguously tiny (or nonexistent) effects. Substantial reliance on observational data for which causal inference is notoriously difficult also limits the clarifying ability of nutrition science. When the data are not clear, opinions and conflicts of interest both financial and nonfinancial may influence research articles, editorials, guidelines, and laws. Therefore, disclosure policies are an important safeguard to help identify potential bias. “
While the potential for financial conflict in relationship to the food industry is well recognised and there are now well-established “disclosure norms”, other conflicts, of which there are many, are not routinely acknowledged, let alone, disclosed.
For one, there are significant financial conflicts that have nothing to do with taking money from industry:
“Many nutrition scientists and experts write books about their opinions and diet preferences. Given the interest of the public in this topic, books about nutrition, diets, and weight loss often appear on best-selling lists, even though most offer little to no evidence to support their frequently bold claims.”
Furthermore,
“Financial conflicts of interest can also appear in unexpected places. For example, many not-for-profit nutrition initiatives require considerable donor money to stay solvent. Public visibility through the scientific literature and its reverberation through press releases, other media coverage, and social media magnification can be critical in this regard.”
Even these financial conflicts can perhaps be dealt with through established disclosure norms.
But conflicts can get even more complicated when it starts reflecting researchers’ own personal views and biases::
“Allegiance bias and preference for favorite theories are prevalent across science and can affect any field of study. It is almost unavoidable that a scientist eventually will form some opinion that goes beyond the data, and they should. Scientists are likely to defend their work, their own discoveries, and the theories that they proposed or espoused.”
While that is certainly true for any area of research,
“Nutrition scientists are faced with an additional challenge. Every day they must make numerous choices about what to eat while not allowing those choices to affect their research. Most of them also have been exposed to various dietary norms from their family, culture, or religion. These norms can sometimes be intertwined with core values, absolutist metaphysical beliefs, or both. For instance, could an author who is strongly adherent to some religion conclude that a diet-related prescription of his or her religion is so unhealthy as not to be worthwhile?”
Moreover,
“Advocacy and activism have become larger aspects of the work done by many nutrition researchers, and also should be viewed as conflicts of interest that need to be disclosed. These endeavors often spring from some of the noblest intentions and can lead to invaluable contributions to society and public health in particular. However, advocacy and activism are also orthogonal to a key aspect of the scientific method, which is to not take sides preemptively or based on belief or partisanship. Examples of white-hat bias (bias that distorts scientific evidence in support of a perceived righteous end such as better human health) have been reported.”
The authors therefore propose that,
“…it is important for nutrition researchers to disclose their advocacy or activist work as well as their dietary preferences if any are relevant to what is presented and discussed in their articles. This is even more important for dietary preferences that are specific, circumscribed, and adhered to strongly. For example, readers should know if an author is strongly adherent to a vegan diet, the Atkins diet, a gluten-free diet, a high animal protein diet, specific brands of supplements, and so forth if these dietary choices are discussed in an article. The types of articles in which relevant disclosure should be expected include original research, reviews, and opinion pieces (such as editorials).”
As with financial disclosures,
“Such disclosure should not be seen as an admission of lack of integrity. To the contrary, disclosure strengthens the perceived integrity of the author. Moreover, some disclosures may end up being advantageous depending on future research findings. For example, if at some point strict vegan diets are shown definitively to confer unmatched health benefits, an author who previously disclosed strong adherence to that diet may receive extra recognition and acclaim for his or her prescient wisdom….Availability of these disclosures would allow readers to be either more skeptical or more inspired (depending on how they view the presented evidence and arguments).”
Although the article focuses on nutrition research, the authors acknowledge that similar biases may exist in other areas of research. In my own experience, “ideological biases” (although well-intended) are pervasive through much of the research and publications on topics ranging from physical activity to public health, where I often see strong recommendations made based on evidence that is not even remotely as robust or rigorous as the evidence that comes from, say a large randomised clinical trials of a new prescription drug.
I certainly agree with the authors’ recommendation that,
“As a general rule, if an author’s living example could be reasonably expected to influence how some readers perceive an article, disclosure should be encouraged. Authors who have strong beliefs and make highly committed choices for diet or other behaviors should not hesitate to disclose them. Doing so may help everyone understand who is promoting what and why.”
@DrSharma
Edmonton, AB
Surviving Cancer Doesn’t Make You An Oncologist!
 In my TEDx talk “How to Lose 50 Pounds and Keep Them Off” released earlier this week, I recommend that people struggling with obesity seek help from a health care professional trained in chronic disease management.
In my TEDx talk “How to Lose 50 Pounds and Keep Them Off” released earlier this week, I recommend that people struggling with obesity seek help from a health care professional trained in chronic disease management.
Given that most of what we deal with in medicine is chronic disease, one would imagine that almost any health professional can give competent advice on living with a chronic disease.
Unfortunately, while this may be true for diabetes or hypertension or most of the other common chronic diseases that virtually all health professionals at some time in their careers will have learnt to manage, very few will have any experience at all in treating obesity – let alone even thinking of obesity as a chronic disease.
It is therefore not surprising that when it comes to weight management people often turn to lay “experts” who proclaim themselves as “experts” simply because they have somehow managed to lose weight and are (at least for now) managing to keep it off.
Often, these “experts” (not seldom with the help of a savvy publicist) will launch a lucrative speaking business including authoring self-help books, offering diet/exercise plans, appearing on TV talk shows, endorsing supplements or promoting whatever it is they believe helped them lose their weight.
Unfortunately, as I hear over and over from my patients, these self-proclaimed “experts” who, having “conquered” their obesity, have now embarked on a mission to help you “conquer” yours, are about as effective as any other commercial fad diet.
This is because, we have long known that in obesity, what works for one person may be entirely ineffective in the next.
Just because you lost weight by following (insert your approach here), does not mean that you have found the Holy Grail of weight management and are now somehow professionally qualified to offer your help to everyone else.
Living with heart disease does not make you cardiologist!
Surviving cancer does not make you an oncologist!
Losing weight does not make you an obesity specialist!
This is not to say that the “patient” experience is not important or that “peer support” cannot be a valuable tool in chronic disease management.
Indeed many successful weight management programs including Taking Off Pounds Sensibly or Weight Watchers rely heavily on “peer support”, in that groups are generally run by lay people who are themselves “affected”.
While this makes a lot of sense, it does not replace the need for a professionally qualified health professional to ensure that all goes well.
Just because you are a member of the local diabetes support group does not mean you no longer need to meet with your diabetes educator or family doctor.
Just because your dearest friend survived a heart attack and is willing to share her experience and support, doesn’t mean you no longer need to see a cardiologist for your own heart problems.
This is an important distinction and should be a warning to not rely too heavily on people peddling their “success stories”.
While it may well of interest to listen and perhaps even examine their suggestions, hoping that what worked for them will work for you, is both shortsighted and can potentially do more harm than good.
Do I wish that all health professionals had a sound understanding of obesity management and are there to support people living with obesity – of course I do.
But I know we are not there yet and it will take time before health professionals begin seeing their role change from simply “recommending weight loss” t0 actually having the expertise and experience required to help patients better manage their weight and health.
@DrSharma
Edmonton, AB
Welcome To The International Congress on Obesity, Vancouver 2016
 This weekend sees the start of the XIII International Congress on Obesity (ICO), hosted by the World Obesity Federation in partnership with the Canadian Obesity Network (CON) in Vancouver, Canada.
This weekend sees the start of the XIII International Congress on Obesity (ICO), hosted by the World Obesity Federation in partnership with the Canadian Obesity Network (CON) in Vancouver, Canada.
As this year’s Congress President, together with World Obesity Federation President Dr. Walmir Coutinho, it will be our pleasure to welcome delegates from around the world to what I am certain will be a most exciting and memorable event in one of the world’s most beautiful and livable cities.
The program committee, under the excellent leadership of Dr. Paul Trayhurn, has assembled a broad and stimulating program featuring the latest in obesity research ranging from basic science to prevention and management.
I can also attest to the fact that the committed staff both at the World Obesity Federation and the Canadian Obesity Network have put in countless hours to ensure that delegates have a smooth and stimulating conference.
The scientific program is divided into six tracks:
Track 1: From genes to cells
- For example: genetics, metagenomics, epigenetics, regulation of mRNA and non–coding RNA, inflammation, lipids, mitochondria and cellular organelles, stem cells, signal transduction, white, brite and brown adipocytes
Track 2: From cells to integrative biology
- For example: neurobiology, appetite and feeding, energy balance, thermogenesis, inflammation and immunity, adipokines, hormones, circadian rhythms, crosstalk, nutrient sensing, signal transduction, tissue plasticity, fetal programming, metabolism, gut microbiome
Track 3: Determinants, assessments and consequences
- For example: assessment and measurement issues, nutrition, physical activity, modifiable risk behaviours, sleep, DoHAD, gut microbiome, Healthy obese, gender differences, biomarkers, body composition, fat distribution, diabetes, cancer, NAFLD, OSA, cardiovascular disease, osteoarthritis, mental health, stigma
Track 4: Clinical management
- For example: diet, exercise, behaviour therapies, psychology, sleep, VLEDs, pharmacotherapy, multidisciplinary therapy, bariatric surgery, new devices, e-technology, biomarkers, cost effectiveness, health services delivery, equity, personalised medicine
Track 5: Populations and population health
- For example: equity, pre natal and early nutrition, epidemiology, inequalities, marketing, workplace, school, role of industry, social determinants, population assessments, regional and ethnic differences, built environment, food environment, economics
Track 6: Actions, interventions and policies
- For example: health promotion, primary prevention, interventions in different settings, health systems and services, e-technology, marketing, economics (pricing, taxation, distribution, subsidy), environmental issues, government actions, stakeholder and industry issues, ethical issues
I look forward to welcoming my friends and colleagues from around the world to what will be a very busy couple of days.
For more information on the International Congress on Obesity click here
For more information on the World Obesity Federation click here
For more information on the Canadian Obesity Network click here
@DrSharma
Edmonton, AB
How To Remove A Gastric Band
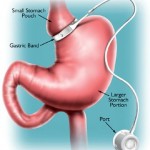 When adjustable gastric banding was first introduced as a treatment for severe obesity, it was touted not only as a surgically simple procedure (which it is) but also as having the advantage of being “reversible”.
When adjustable gastric banding was first introduced as a treatment for severe obesity, it was touted not only as a surgically simple procedure (which it is) but also as having the advantage of being “reversible”.
Unfortunately, as many patients who have had bands implanted only to discover that they either did not work or created all kinds of other problems, “simply” removing the band was often anything but “simple”.
Moreover, as with any obesity treatment, removing the band (or in other words, stopping the treatment) virtually guarantees weight regain, leaving the patients often back to where they were (if not worse off) than before the operation.
In reality this means that when bands need to be removed, they often have to be followed by a more definitive procedure like a sleeve gastrectomy (SG) or a Roux-en-Y gastric bypass (RYGB) (the operations that should probably have been done in the first place).
The debate that continues to rage between surgeons, is whether or not it is safer to remove the band and do the more definitive operation within a single procedure or in two separate operations.
Now my colleague Jerry Dang from the University of Alberta, Edmonton, Canada, has published a systematic review and meta-analysis of the two approaches in Obesity Surgery.
According to their analysis of 11 studies that included 1370 patients, there was no difference in the rates of complications, morbidity, or mortality between one-step and two-step revisions for both RYGB and SG groups.
Thus, the authors conclude that both immediate or delayed revisional bariatric surgeries are reasonable and comparable options for band revisions.
Overall however, this should not distract from the fact that a band removal in itself can be a difficult operation with its own set of complications and given the number of bands that have to be removed, it may be best be recommended that preference be given to doing an SG or RYGB as the primary procedure.
Fortunately, this is indeed proving to be the practice at an increasing number of surgical centres.
@DrSharma
Edmonton, AB


