Low Carb No Better Than High Protein For Weight Loss
 When I finished medical school in 1986, restricting protein intake in patients with reduced kidney function was still part of standard practice. This practice, has since been long abandoned (having probably done more harm than good), but notions about higher protein intake somehow being harmful to kidneys lingers on.
When I finished medical school in 1986, restricting protein intake in patients with reduced kidney function was still part of standard practice. This practice, has since been long abandoned (having probably done more harm than good), but notions about higher protein intake somehow being harmful to kidneys lingers on.
Thus, I was interested by a recent study by Pedersen and colleagues from the University of Adelaide, Australia, published in Nutrition, Metabolism and Cardiovascular Disease, which examined the effect of of a high-protein calorie restricted diet on overweight individuals with type 2 diabetes, albuminuria and reduced kidney function (estimated GFR of >40 ml/min/1.73m2).
Subjects were randomised to 12 months of a standard-protein diet (protien:fat:carbohydrate ratio 20:30:50 % of energy) and one where energy from carbohydrates was reduced and replaced by protein (30:30:40 % of energy).
Both groups lost a significant amount of weight over 12 months (6.6 Kg on the standard-protein diet, 9.7 Kg on the high-protein diet).
The diets had no impact on kidney function despite improvements in diabetes control.
Of note, only 45 of the 76 volunteers completed the study – a drop-out rate of over 40%
Overall, the study shows that differences in carb to protein ratios matter neither in terms of weight loss nor in their impact on kidney function.
Perhaps, even more importantly, the study shows that trying to keep people on diets – even in clinical trials – is challenging, with almost half the subjects abandoning their diet within 12 months.
As I have noted before, diets only work when you stick with them. Rather than obsessing about the exact composition of your diet, it may be best to chose the one you like best and can actually stay on.
@DrSharma
Edmonton, AB
Peritoneal and Exit Site Infections in Obese Patients on Peritoneal Dialysis
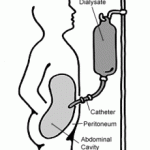 Given my background in nephrology, a recent paper by Sharon Nessim and colleagues from the Jewish General Hospital, Montreal, Quebec, published in Peritoneal Dialysis International caught my attention.
Given my background in nephrology, a recent paper by Sharon Nessim and colleagues from the Jewish General Hospital, Montreal, Quebec, published in Peritoneal Dialysis International caught my attention.
This paper looked at the relationship between BMI and the risk of peritonitis and catheter infections in about 1000 peritoneal dialysis (PD) patients in the province of Manitoba who received PD during the period 1997 – 2007.
In unadjusted analyses, patients in the highest BMI quartile (median: 33.5) had an increased risk of peritonitis overall, and also an increased risk of peritonitis with gram-positive organisms and coagulase-negative Staphylococcus (CNS).
However, after multivariate adjustment for age, sex, diabetes, cause of renal disease, Aboriginal race, PD modality, and S. aureus nasal carriage, the relationship between overall peritonitis risk and BMI disappeared, but the increased risk of CNS peritonitis among patients in the highest BMI quartile persisted (hazard ratio: 1.80).
Thus, while patients with higher BMI on PD are clearly at greater risk of experiencing infections, this risk is largely explained by other concomitant factors than by BMI itself. If anything, obese patients in the highest weight category appear more prone to CNS peritonitis.
Obviously this risk is certainly not high enough or specific enough to deter anyone from chosing PD as a mode of dialysis. Nevertheless, it is probably a finding that clinicians (especially nephrologists) may need to be aware of.
AMS
Edmonton, Alberta
![]() Nessim SJ, Komenda P, Rigatto C, Verrelli M, & Sood MM (2012). FREQUENCY AND MICROBIOLOGY OF PERITONITIS AND EXIT-SITE INFECTION AMONG OBESE PERITONEAL DIALYSIS PATIENTS. Peritoneal dialysis international : journal of the International Society for Peritoneal Dialysis PMID: 22942268
Nessim SJ, Komenda P, Rigatto C, Verrelli M, & Sood MM (2012). FREQUENCY AND MICROBIOLOGY OF PERITONITIS AND EXIT-SITE INFECTION AMONG OBESE PERITONEAL DIALYSIS PATIENTS. Peritoneal dialysis international : journal of the International Society for Peritoneal Dialysis PMID: 22942268
.
Hindsight: Obesity As a Risk Factor In Kidney Transplant Recipients
Continuing my review of past papers on obesity, here is one that I am particularly fond of, as there was rather limited information on this topic at the time – namely the impact of obesity on kidney transplant recipientsThe paper, published in Nephrology, Dialysis, & Transplantation in 2001, was authored by my doctoral student Tobias Pischon, who is now Professor for Molecular Epidemiology at the Charité Berlin and Head of the Molecular Epidemiology Group of the Max-Delbrück-Center for Molecular Medicine (MDC) Berlin-Buch.
In our systematic review, we identified nine relevant studies with patient follow‐up ranging from 24 to 120 months in a total of 2791 kidney transplant patients.
As could perhaps be expected, obese transplant recipients were more likely to experience a wide range of problem including systematic and wound infections, gastrointestinal problems, wound complications, admission to intensive care for cardiac and pulmonary complications, urological problems, new-onset diabetes, hypertension, phlebitis, and leg ulcers.
While obesity did not appear to affect acute rejection, there were some reports on delayed graft function possibly related to longer operation times or urological complications.
Virtually all investigators reported a somewhat (but not dramatically) lower graft survival in obese compared with non‐obese renal transplant recipients, with differences appearing as early as 1 year post-transplantation. According to one report, obese patients were also more likely to die earlier even with a functioning graft.
We also found consistent reports on marked increase in body weight post transplant (often a side effect of steroid medication) with obese patients often gaining more weight than normal weight patients resulting in more diabetes and cardiac problems.
From these findings we concluded that:
“Obesity is commonly present in patients undergoing renal transplantation and is significantly associated with a higher overall mortality and reduced allograft survival. Obese patients experience a greater incidence of perioperative complications, often resulting in delayed graft function, early graft loss and, in some cases, delayed discharge from hospital. In contrast, immunological complications of transplantation are apparently not related to body weight. A substantial proportion of transplant patients experience a marked increase in body weight following transplantation and obesity remains a significant cause of diabetes and cardiac complications in this population.”
In the past 10 years, with the continuing increase in obesity, both pre- and post-transplant obesity remains a significant issue in patients with end stage kidney failure.
According to Google Scholar, this paper has been cited 46 times.
AMS
Canmore, Alberta
Obesity Related Kidney Disease
Last week, I gave a talk on obesity assessment and management at the Nephrological Seminar in Heidelberg, Germany.This annual conference, now in its 35th year, is hosted by Eberhard Ritz, who I have known for most of my professional career. Ritz, is certainly one of Germany’s pre-eminent nephrologists and as an emiritus professor still appears as active as ever, churning out article after article on a remarkably wide range of topics in nephrology and hypertension.
At this year’s Seminar, Ritz also presented an overview of the impact of obesity on kidney function. The summary of this talk was recently published in Current Opinions in Nephrology and Hypertension.
As Ritz points out, obesity has now been show to be a risk factor for chronic kidney disease, independent of its common association with diabetes and/or hypertension.
The earliest record of nephrotic range proteinuria (where patients excrete several grams of protein with their urine every day) in patients with severe obesity, was in 1974 by Weisinger. This report described four patients with severe proteinuria, which decreased with weight loss and reappeared with subsequent weight regain.
Weisinger also described a typical focal segmental glomerulosclerosis (FSGS) on histological exams of kidney biopsies from these patients, findings that appeared quite different from other causes of proteinuria (e.g. diabetic nephropathy).
Subsequent studies have confirmed similar findings in other patients with severe obesity and protienuria and between 1986 and 2000 a more than 10-fold increase in the prevalence of this problem was reported.
Also, more recently, similar (albeit less severe) FSGS has been reported even with moderate obesity as well as in obese children and adolescents.
Although proteinuria is generally reduced with weight loss, few patients manage to keep the weight off, resulting in recurrence of protein excretion and deterioration of renal function.
More recently, bariatric surgery, which generally results in much better long-term weight loss, has been reported to reduce proteinuria and stabilse renal function in obese patients with FSGS.
Of course, although FSGS appears to be the lesion that is most typically associated with obesity, it must be noted that all forms of kidney disease can be worsened by excess weight.
Unfortunately, large prospective trials of weight loss (surgical or non-surgical) to prevent the progressive loss of kidney function in patients with excess weight are lacking.
Such studies may be particularly relevant today, as type 2 diabetes, a condition that often goes into prolonged remission after bariatric surgery, is now emerging as the single most common driver of endstage kidney failure leaving patients with no option other than dialysis or kidney transplantation.
I am sure that many obese patients with progressive renal failure would likely prefer bariatric surgery to a life on dialysis – but whether or not this is indeed the best option will probably first have to be shown in a trial.
Nevertheless, I am sure that some of my readers will probably know of cases where renal function (protein excretion and/or loss of filtration rate) was affected by weight loss – I’d certainly be very interested in hearing about these observations.
AMS
Edmonton, Alberta
Ritz E, Koleganova N, & Piecha G (2011). Is there an obesity-metabolic syndrome related glomerulopathy? Current opinion in nephrology and hypertension, 20 (1), 44-9 PMID: 21088574
The Kidney as a Target for Diabetes Treatment?
This week, I am attending the 46th European Association for the Study of Diabetes (EASD) Annual Meeting in Stockholm, Sweden, where there is considerable enthusiasm about the many new drugs and drug classes for diabetes treatment, which are likely to hit the market in the next few years.
One such group of novel orally active anti-diabetic agents are the sodium-glucose cotransporter 2 (SGLT2) inhibitors (gliflozins), which act by blocking this transporter in the renal proximal tubule.
As reader may know, blood glucose is freely filtered in the kidney and is normally entirely reabsorbed in the renal tubule (which is why there is usually almost no glucose in the urine of non-diabetic individuals).
Experts may care that SGLT2 is a low-affinity high-capacity transporter sodium/glucose co-transporter, in contrast to SGLT1, located later in the tubule, which is a high-affinity low-capacity transporter and normally reabsorbs any remaining glucose missed by SGLT2.
As outlined by Clifford Bailey from Aston University, Birmingham, UK, the history of SGLT2 inhibitors dates as far back as 1835, when phlorizin, a naturally occurring flavonoid, later shown to be a competitive inhibitor of renal glucose transport, was identified in the bark of the apple tree.
Phlorizin’s glycosuric effect was reported in the 1860s, its renal actions in rat kidneys was shown in 1903 and in humans in 1933. Subsequently, it was shown that phlorizin may well have a significant glucose-lowering effects in patients with diabetes.
However, because phlorizin inhibits both SGLT2 and SGLT1 it had to be pharmacologically tweaked to make it specific for SGLT2 but also to make it longer acting. SGLT2 inhibitors that are currently in development, include dapagliflozin (AstraZeneca), canagliflozin (Johnson & Johnson), sergliflozin (GSK), ASP1941 (Astellas), BI 10773 (Boehringer Ingelheim), and LX4211 (Lexicon).
At blood glucose levels of 8 mmol/L, about 260 g (or almost half a pound of glucose) is filtered every day.
SGLT2 inhibitors reduce glucose reabsorption by about 25%, which means that about 70g of glucose or 280 calories are lost in the urine per day; the higher the blood glucose levels, the greater the caloric loss.
To put this in perspective, if the 280 calories lost in the urine are not replaced by dietary energy intake (in any form), weekly caloric deficit would be around 2000 Kcal, theoretically resulting in about 0.75 lbs weight loss.
Thus, although the primary objective of SGLT2 inhibitors is to help improve blood glucose levels in diabetic patients (which they do), an interesting “side effect” of this treatment is modest weight loss (2-3.5%).
The SGLT2 inhibitors appear to be well tolerated with almost no renal side effects but perhaps a minor increase in genital infection in women.
Numerous clinical trials (many presented at this conference) show that the gliflozins can be combined with both oral and injectable anti-diabetic agents and generally result in relevant improvements in glycemic control.
As a nephologist, who now works mainly in the area of obesity and metabolic diseases, I am particularly intrigued by the mode of action of these compounds and the fact that this novel class of agents specifically targets the kidney.
The fact that gliflozins also, albeit modestly, promote weight loss, makes these particularly attractive for my patients, as most other oral agents (except metformin and DPP IV inhibitors) tend to promote clinically significant weight gain.
AMS
Stockholm, Sweden
You can now also follow me and post your comments on Facebook
Vallon V, & Sharma K (2010). Sodium-glucose transport: role in diabetes mellitus and potential clinical implications. Current opinion in nephrology and hypertension, 19 (5), 425-31 PMID: 20539226





