Does Bariatric Surgery Induce Long-Term Changes in Gut Bugs That Promote Weight Loss?
 With all the talk about how gut bugs may play a key role in susceptibility to weight gain (and resistance to weight loss?), we are learning much about this issue from animal studies.
With all the talk about how gut bugs may play a key role in susceptibility to weight gain (and resistance to weight loss?), we are learning much about this issue from animal studies.
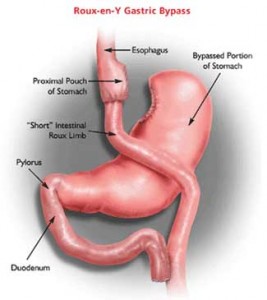
However, now a human study by Valentina Tremaroli and colleagues published in Cell Metabolism, not only shows that bariatric surgery can induce distinct changes in gut bacteria but also that transferring stool from these individuals to germ-free mice changes their metabolism towards greater utilization of carbohydrates for fuel.
As a first step, the researchers examined the gut microbiota of 14 weight-stable women 9 years after randomization to either gastric bypass (n=7) or vertical banded gastroplasty (n=7) and matched for weight and fat mass loss.
As a control, they also analyzed the gut microbiota of two groups of non-operated women with a BMI matched to the patients’ pre-surgical BMI (OBS) and post-surgical BMI (Ob).
The gut bacteriome was significantly altered in both surgical groups with some difference between the two procedures: changes in the gastric-bypass patients suggest an increased energy flux into sugar metabolism and glycolysis, whereas the VBG patients showed an enrichment of pathways for amino acid uptake and metabolism and for glyoxylate metabolism pointed to the utilization of amino acids and acetate for energy production.
There were also important difference in bile acid metabolism in the surgery groups.
The authors are comfortable that these changes result from bariatric surgery and are not related to BMI per se, since the OBS and Ob microbiomes were similar. Also, similar changes in gut microbiota have not been seen during dietary interventions for weight loss.
Thus, the authors note that,
“…our results suggest that bariatric surgery produces a specific shift in the microbiota that persists for up to a decade after surgery and is different from shifts related to dietary interventions for weight loss.”
In a second set of experiments, the researchers transplanted the microbiome from the various groups to germ free mice.
Mice colonized with gastric-bypass and banding microbiota for 2 weeks accumulated 43% and 26% less body fat, respectively, than mice colonized with OBS microbiota despite the fact that body weight gain and food intake did not differ between the groups during the 2-week colonization period.
In addition, metabolic studies showed decreased utilization of carbohydrates and increased utilization of lipids as fuel in recipients of gastric-bypass microbiota.
Thus,
“…this study clearly shows that bariatric surgery has long-term effects on the composition and functional capacity of the gut microbiota and that these changes have the potential to modulate host metabolic regulation, thus adding evidence for the transmissibility of the human adiposity phenotype through the gut microbiota.”
“Our results also show that two different bariatric surgery procedures, namely RYGB and VBG, have similar long-term effects on the gut microbiome in women matched for BMI and fat mass loss. However, the two bariatric surgery procedures might result in different functionality due to different intestinal environmental conditions…. Importantly, the changes in the microbiome were not dependent on BMI or degree of weight and fat mass loss, thus revealing shifts in the gut microbiota that were specific to bariatric surgery.”
These findings should certainly do away with the simplistic notions of how and why bariatric surgery works – perhaps it is high time we discarded the notions of “restrictive” or “malabsorptive” surgery.
@DrSharma
Edmonton, AB
Can Dietary Emulsifiers Promote Obesity?
 Emulsifiers are amongst the most commonly used food additives to improve the texture and palatability of foods ranging from margarine and mayonnaise to ice cream and baked goods.
Emulsifiers are amongst the most commonly used food additives to improve the texture and palatability of foods ranging from margarine and mayonnaise to ice cream and baked goods.
Now a study by Benoit Chassaing and colleagues published in NATURE, suggests that dietary emulsifiers may promote weight gain and the metabolic syndrome by altering the composition of intestinal microbes.
The researchers hypothesized that emulsifiers may increase bacterial translocation across intestinal mucosa, thereby promoting local and systemic inflammation as well as affecting the composition of gut bacteria.
Their study in mice show that relatively low concentrations of two commonly used emulsifiers (carboxymethylcellulose and polysorbate-80) can induce low-grade systemic inflammation, weight gain and features of the metabolic syndrome, as well as promote intestinal inflammation in mice susceptible to inflammatory bowel disease.
Importantly, they used germ-free mice and faecal transplants to show that these changes can be induced simply by transferring the gut microbes from emulsifier-treated animals to controls.
As the authors note,
“These results support the emerging concept that perturbed host-microbiota interactions resulting in low-grade inflammation can promote adiposity and its associated metabolic effects. Moreover, they suggest that the broad use of emulsifying agents might be contributing to an increased societal incidence of obesity/metabolic syndrome and other chronic inflammatory diseases.”
While these findings (if replicated in humans) certainly point to the industrial use of food emulsifiers as a potential cause of the global increase in obesity and inflammatory bowel disease, given that these compounds are present in virtually all processed foods, they may well be difficult to avoid.
Guess it’s back to home cooking with raw ingredients.
@DrSharma
Edmonton, AB
City Sewage Predicts Obesity Rates?
 And if you thought obesity news could not get quirkier – how about this one?
And if you thought obesity news could not get quirkier – how about this one?
A study by Ryan Newton and colleagues in mBio, the open access journal of the American Society for Microbiology, found that the bacterial composition of city sewage can almost precisely predict obesity rates in that city.
The researchers studied the microbial community of sewage from 71 US cities from 31 states using high-througput 165 rRNA gene sequencing technology.
Although on average only 15% of bacterial sequences in each sample represented bacteria known to occur in human stool, they were able to capture most (97%) of human fecal oligotypes.
Based on the distribution of three primary oligotypes representing different proportions of Bacteroidaceae, Prevotellaceae, or Lachnospiraceae/Ruminococcaceae, the researchers were able to predict whether samples came for cities with high or low prevalence of obesity with 81-89% accuracy.
No such relationship was found with non-fecal oligotypes, suggesting that this relationship was indeed due to the representation of human fecal bacteria in the sewage samples.
Obviously, it is very possible that the sewage bacterial composition reflects “lifestyles” associated with obesity rather than actual body weights, but the very fact that it was possible to identify important predictive differences in bacterial patterns between cities with varying obesity rates, together with the increasing recognition that gut bacteria may well play a role in obesity (and other metabolic diseases), is fascinating enough.
Should these findings be reproducible across other populations, I can only wonder whether sewage sampling may one day serve as a simple way to study changes in nutrition and obesity rates in whole populations.
Indeed, I can picture future public health scientists poring over sewage data to check if their public health policies to reduce obesity are in fact working.
@DrSharma
Edmonton, AB
Weight Gain After Fecal Transplant
 Regular readers will be well aware of the emerging science of the human microbiome, suggesting that the several pounds of bacteria that live in our gut may well play an important role in the development of obesity and other metabolic diseases.
Regular readers will be well aware of the emerging science of the human microbiome, suggesting that the several pounds of bacteria that live in our gut may well play an important role in the development of obesity and other metabolic diseases.
Readers may also have heard of the use of fecal transplants, i.e. the transfer of stool samples from a healthy donor, to treat patients suffering from a severe form of diarrhoea caused by Clostridium difficile.
Now an interesting report by Neha Alang and Colleen Kelly from Providence, Rhode Island, published in Open Forum Infectious Diseases, describes a case of remarkable weight gain in a non-obese woman following the successful treatment of her Clostridium difficile infection with a fecal transplant from her overweight daughter.
Prior to undergoing the fecal transplant in 2011, the 32 year old patient, who had always been of normal weight, was weight stable at 136 pounds (BMI 26).
Her stool “donor” was her overweight but otherwise healthy teenage daughter.
Sixteen months after fully recovering from her C. diff infection, the patient weighed 170 pounds (BMI 33).
Despite continuing efforts to lose weight with diet and exercise, she now weighs 177 pounds (BMI 34.5).
Despite these remarkable development, it is important not to jump to conclusions.
Not only is this just a single case report, but the authors also discuss other possible explanations including treatment with antibiotics for Helicobacter pylori infection and the stress related to illness.
Nevertheless, given that the patient had never been overweight before, and we do have an increasing number of studies suggesting that the bacteriome may indeed play a role in obesity, this report should give some food for thought.
If nothing else, it may be prudent to avoid overweight or obese donors when considering stool transplants for C. diff infections.
@DrSharma
Edmonton, AB
Can Gut Bugs Cause Depression?
 Regular readers will by now be well aware of the rapidly growing body of researcher supporting the idea that your gut bugs (of which you have more than you have cells in your body) may well play a key role in determining your risk for obesity and other metabolic disorders.
Regular readers will by now be well aware of the rapidly growing body of researcher supporting the idea that your gut bugs (of which you have more than you have cells in your body) may well play a key role in determining your risk for obesity and other metabolic disorders.
Now, a paper by Ruth Ann Luna and Jane Foster from Baylor College of Medicine and McMaster University, respectively, published in Current Opinion in Biotechnology, review the evidence that gut bugs may well also have significant effects on your stress response as well as other aspects of mental healthy, including depression.
As one example, they cite a study that shows,
“…a general underrepresentation of the Bacteroidetes phylum in depressed patients and an association of the Lachnospiraceae family with the depression group, and interestingly, even with a decrease in Bacteroidetes, specific operational taxonomic units (OTUs) identified as members of the Bacteroidetes phylum correlated with depression.”
They also cite a number of studies showing that stress can affect gut bug populations and that certain gut bacteriomes are associated with a greater stress response, suggesting that the relationship between gut bugs and stressors may well be a two-way street.
The authors go on to describe a number of pathways that may link gut bugs to humoral, neural, and cellular signaling pathways to brain function.
Clearly, this appears a rich area of research that may well reveal pathways common to both neurological and metabolic issues, both of which may turn out to be amenable to dietary and probiotic interventions.
@DrSharma
London, UK
![]() Luna RA, & Foster JA (2014). Gut brain axis: diet microbiota interactions and implications for modulation of anxiety and depression. Current opinion in biotechnology, 32C, 35-41 PMID: 25448230
Luna RA, & Foster JA (2014). Gut brain axis: diet microbiota interactions and implications for modulation of anxiety and depression. Current opinion in biotechnology, 32C, 35-41 PMID: 25448230


