Obesity Management: Handguns versus Slingshots?
Tuesday, December 7, 2010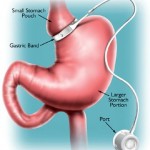 Regular readers will be well aware that for most people with severe obesity, bariatric surgery is currently the only therapeutic option shown to reduce both long-term morbidity and mortality.
Regular readers will be well aware that for most people with severe obesity, bariatric surgery is currently the only therapeutic option shown to reduce both long-term morbidity and mortality.
But this may now change – as yesterday, an advisory panel appointed by the US Food and Drug Administration (FDA), voted 8-2 to extend the currently approved use of the LAP-BAND® System for adults with a BMI of at least 35 or a BMI ≥30 and at least one comorbid condition.
This means that bariatric surgery is about to venture into the area of less-severe obesity, in fact, as a treatment option for people with a BMI as low as 30, provided they have weight-related comorbidities
The Committee’s decision was based, among other considerations, on the results of a 12-month prospective, single-arm, non-randomized, multi-center five-year study involving 149 moderately obese patients, who on average had been obese for 17 years.
In this study, 83.9% of the patients lost at least 30% of their excess weight at the one-year timepoint, more than twice the percentage required for success by the FDA. Remarkably, more than 65% of the patients in the trial were no longer obese after one year.
This degree of weight loss was accompanied by a substantial improvement in dyslipidemia, Type 2 diabetes, and hypertension as well as improvement in Quality of Life (QoL).
During the 12-month study period, the types of Adverse Events (AEs) reported by patients were as expected for the surgical procedure, such as vomiting, dysphagia, and gastroesophageal reflux disease (GERD). Most AEs were mild to moderate in severity and resolved in less than 4 weeks.
Readers may well recall previous posts on the significant impact of adjustable gastric banding in patients with type 2 diabetes but also on the remarkable peri-operative safety of this type of surgery.
Nevertheless, for me as a non-surgeon, this recommendation in favour of bariatric surgery (albeit in this case, specifically for adjustable gastric banding), as a safe and effective treatment for mild to moderate obesity, stands in remarkable contrast to the much tougher stance recently taken by the FDA on pharmacological treatment options for obesity.
Thus, readers will recall that the FDA recently not only took a surprisingly negative view of lorcaserin and qnexa, but also decided to remove sibutramine from the market, based on putative cardiovascular safety concerns seen in high-risk individuals for whom this drug was never intended.
Interestingly, today, the FDA will also hear from an advisory panel on another obesity combination drug (contrave) and I will be curious to see whether or not the advisors find the data on this compound convincing enough for a strong positive vote in its favour.
Although, I certainly agree that gastric banding can provide a useful treatment option for moderate obesity, it is hard to see how the risks inherent in undergoing surgery (even under the safest circumstances) can even begin to compare with the relative safety of taking a pharmacological agent – especially, as in the case of contrave, one that consists of a combination of compounds (i.e. naltrexone SR/bupropion SR), that have been on the market for over 20 years for other indications.
One can only wonder about the curious rationale behind the FDA’s decisions to approve a surgical device for treating mild to moderate obesity, while making it exceedingly difficult, if not impossible, to provide the very same patients with pharmacological options to manage their excess weight.
Interestingly, with the approval of the LAP-BAND for use in patients with a BMI as low as 30, we may now wonder if drug companies will perhaps need to go back to the drawing board and not only prove that their drugs are as effective but also as safe as having your stomach surgically constrained.
Somehow I cannot quite dispel the mental picture of one arm of the FDA happily approving the use of a handgun to go after vermin while another arm of the same agency strictly prohibits the use of a slingshot, because the latter may just be too dangerous to be used for the very same purpose.
Truly interesting times for obesity management.
AMS
Ottawa, Ontario
Disclaimer: I have received consulting and speaking honoraria from Allergan, the makers of the LAP-BAND as well as consulting honoraria from the makers of the pharmaceutical agents mentioned in this post.


