Conversation Cards for Adolescents© – Helping adolescents make healthy lifestyle changes

Today’s post comes from Maryam Kebbe, a fourth year Doctoral student studying under the supervision of Dr. Geoff Ball in the Department of Pediatrics at the University of Alberta. Maryam has a passion for patient-oriented obesity research in children and plans to continue doing health research in this area after completion of her PhD.
As readers are probably well aware, often times, the first line of treatment for adolescents seeking health services for obesity management consists of behavioural changes targeting nutrition, physical and sedentary activities, and sleep habits, including an addressing possible issues of mental health.
However, health professionals often encounter a lack of adherence to a healthy behaviours by adolescents with overweight or obesity, resulting in challenges in maintaining or losing weight. This may be due to a number of factors, including difficulties in changing established habits and a lack of consideration for adolescents’ priorities in managing weight. To help with clinical consultations, both adolescents and health professionals can benefit from tools and resources that can be tailored to adolescents with obesity attempting to change their lifestyle habits.
Our team conducted a multi-phase project that included adolescents, health professionals, and researchers to develop Conversation Cards for Adolescents (CCAs), an adolescent-tailored, bilingual (English and French) clinical tool aimed at streamlining conversations and facilitating lifestyle behavior change in adolescents via collaborative goal- setting. Specifically, we completed a review of the literature (1) and a qualitative study including in-person interviews, focus groups, and patient engagement panels (2-5) from which we identified 153 factors that help, may help, or deter adolescents with obesity from adopting healthy lifestyle behaviors. Next, we asked adolescents to prioritize (online survey) and validate (telephone consultations) these factors to help refine our tool (6). The design of this tool included another three rounds of refinements with The Burke Group in collaboration with Obesity Canada.
CCAs comprise a deck of 45 cards. Each card contains an individual statement pertaining to a barrier, enabler, or potential enabler (15 statements per category) that adolescents often encounter in making and maintaining healthy lifestyle changes. These cards are organized across seven categorical suits: nutrition, physical activity, sedentary activity, sleep, mental health, relationships, and clinical factors.
CCAs are intended to be used by adolescents and health professionals and are complementary to an already existing deck of cards (Conversation Cards©) created for parents and health professionals by our research team in 2012 (7-10). Our future steps include completion of a pilot randomized controlled trial to determine the feasibility and user experience of using CCAs by adolescents with obesity and health professionals working in primary care.
Click on the links for more information on CCs and CCAs and order details.
References
- Kebbe M, Damanhoury S, Browne N, Dyson M, McHugh TL, Ball GDC. Barriers to and enablers of healthy lifestyle behaviors of adolescents with obesity: a scoping review and stakeholder consultation. Obesity Reviews, 2017; 12: 1439-1453.
- Kebbe M, Perez A, Buchholz A, Scott S, McHugh TLF, Dyson M, Ball GDC. Health care providers’ delivery of health services for obesity management in adolescents: a multi- centre, qualitative study. BMC Health Services Research, 2019; Under Review.
- Kebbe M, Perez A, Buchholz A, McHugh TLF, Scott SD, Richard C, Dyson MP, Ball GDC. Recommendations of adolescents with obesity to facilitate healthy lifestyle changes: a multi-centre, qualitative study. BMC Pediatrics, 2018; Under Review.
- Kebbe M, Perez A, Buchholz A, McHugh TLF, Scott SD, Richard C, Mohipp C, Dyson MP, Ball GDC. Barriers and enablers for adopting lifestyle behavior changes among adolescents with obesity: a multi-centre, qualitative study. PLoS ONE, 2018; 13: e0209219.
- Kebbe M, Perez A, Buchholz A, Scott SD, McHugh TLF, Richard C, Dyson MP, Ball GDC. Adolescents’ involvement in decision-making for pediatric weight management: a multi-centre qualitative study on perspectives of adolescents and health care providers. Patient Education and Counseling, 2019; In Press.
- Kebbe M, Perez A, Buchholz A, McHugh TLF, Scott SD, Richard C, Dyson MP, Ball GDC. Conversation Cards for Adolescents: a communication and behavior change tool for health care providers and adolescents with obesity. Health Services Research, 2019; Under Review.
- Kebbe M, Byrne J, Damanhoury S, Ball GDC. Following suit: using Conversation Cards for priority-setting in pediatric weight management. Journal of Nutrition Education and Behavior, 2017; 49: 588-592.
- Ball GD, Farnesi BC, Newton AS, Holt NL, Geller J, Sharma AM, Johnson ST, Matteson CL, Finegood DT. Join the conversation! The development and preliminary application of conversation cards in pediatric weight management. Journal of Nutrition Education and Behavior, 2013; 45: 476-478.
- Farnesi BC, Ball GD, Newton AS. Family-health professional relations in pediatric weight management: an integrative review. Pediatric Obesity, 2012; 7: 175-186.
- Farnesi BC, Newton AS, Holt NL, Sharma AM, Ball GD. Exploring collaboration between clinicians and parents to optimize pediatric weight management. Patient Education and Counseling, 2012; 87: 10-17.
The Effects of Obesity on Skeletal Muscle Contractile Function

Skeletal muscle
Given that obesity has profound effects on all organ systems, it is not surprising that excess body fat is also associated with a decrease in muscle function.
The complex biology of the molecular, structural, and functional changes that have been associated with obesity are now extensively discussed in a review article by James Tallin and colleagues, published in the Journal of Experimental Biology.
Without going into the molecular details here, suffice it to say that there is considerable evidence to show and explain why muscular function is impaired in both animal models and humans with excess body fat. (For e.g. at a cellular level, the dominant effects of obesity are disrupted calcium signalling and 5′-adenosine monophosphate-activated protein kinase (AMPK) activity. As a result, there is a shift from slow to fast muscle fibre types. There is also evidence for an impairment in myogenesis resulting from disruption of muscle satellite cell activation. Furthermore, muscle function is affected by insulin resistance and decreased adiponectin levels generally associated with obesity).
Although individuals with obesity will often have a larger muscle mass and may well be stronger than “normal-weight” individuals, when corrected for the amount of extra muscle, it is evident that the muscles are less efficient.
In fact, many of the biochemical and structural changes that occur in obesity are very similar to those found with aging. Not surprisingly, when aging meets obesity, things get even worse.
Although the paper does not discuss the reversibility of these changes with weight loss (or obesity treatment in general), I am aware of other data showing that much of the loss of muscle contractile function associated with obesity can be reversed with weight loss.
A clinical correlate of this is the fact that, following weight loss, individuals often find that it takes far more exercise to burn the same number of calories than before (this is not just because the person is now carrying less weight).
Given the increased recognition that lean body mass is an important determinant of overall health and function, clearly this topic is of continuing interest.
@DrSharma
Edmonton, AB
Applying The Etiological Framework For Obesity Assessment In Clinical Practice
 Continuing with citations from my article in Obesity Reviews on an aeteological framework for assessing obesity, that guides us through a systematic assessment of factors influencing energy metabolism, ingestive behaviour, and physical activity, we ca now apply this framework in clinical practice:
Continuing with citations from my article in Obesity Reviews on an aeteological framework for assessing obesity, that guides us through a systematic assessment of factors influencing energy metabolism, ingestive behaviour, and physical activity, we ca now apply this framework in clinical practice:
This paper provides a comprehensive framework, which should enable clinicians to systematically assess and identify the socio‐cultural, biophysical, psychological and iatrogenic determinants of increased energy intake and reduced energy expenditure in patients presenting with excess weight or weight gain. Beginning with an assessment of energy requirements and metabolism, clinicians should systematically assess the role and determinants of ingestive and activity behaviour to identify the factors promoting positive energy balance. This will enable clinicians to develop management plans that address the root causes of weight gain and move beyond the simplistic and generally ineffective recommendation to ‘eat less and move more’.
Thus for example, in a listless patient ‘self‐medicating’ with food, identification and treatment of depression may be the first step to reducing food intake and preventing further weight gain. In a patient with socioeconomic barriers to healthy eating or physical activity, referral to a social worker who can assist in identifying and accessing community resources may be important. Identification and effective treatment of obstructive sleep apnoea may be the key to increasing activity in someone with this disorder. Psychological counselling to manage alcohol or substance abuse or to help patients deal with binge eating resulting from past trauma, emotional neglect or grief, can put patients on a path to successful weight management. Clearly, the common notion that all forms of obesity can be addressed simply by counselling patients on diet and exercise should be considered ineffective and obsolete.
To conclude this series, we will tomorrow look at some of the potential limitations of this system.
@DrSharma
Edmonton, AB
Factors That Affect Physical Activity
 Continuing with citations from my article in Obesity Reviews on an aeteological framework for assessing obesity, we now turn to the some of the factors that can affect physical activity. Similar to the factors that can affect ingestive behaviour, there are a host of factors that can significantly affect physical activity:
Continuing with citations from my article in Obesity Reviews on an aeteological framework for assessing obesity, we now turn to the some of the factors that can affect physical activity. Similar to the factors that can affect ingestive behaviour, there are a host of factors that can significantly affect physical activity:
Socio‐cultural factors
A wide range of socio‐cultural determinants of physical activity exist. These range from factors related to the built environment (e.g. urban sprawl, walkability, street connectivity), neighbourhood safety, social networks, and public transportation to socioeconomic limitations as well as customs and beliefs that can influence vocational or recreational physical activity. For example, being promoted from a physically active outdoor job to a sedentary indoor job, moving from a dense urban location to a rural or suburban residence, immigration to a Western country, pregnancy and change in familial status or time constraints can all promote sedentariness and increase the risk of weight gain. Indentifying and addressing the socio‐cultural barriers to physical activity can be a key to successful weight management. Patients facing significant socio‐cultural barriers to activity may specifically benefit from counselling by an occupational and/or recreational therapist.
Biomedical factors
Numerous medical conditions can lead to a reduction in or inability to engage in physical activity. These include musculoskeletal pain or immobility resulting from injury, osteoarthritis or fibromyalgia as well as any other condition that can affect physical performance such as cardiorespiratory disease, obstructive sleep apnoea, chronic fatigue, stroke or urinary incontinence. Alleviating these factors and thereby reducing immobility may be the first step in addressing weight management in these patients. Given the predominant role of musculoskeletal disorders and pain as a barrier to mobility and physical activity, these patients may benefit most from physiotherapeutic interventions and pain management.
Psychological factors and mental health
Lack of motivation, low energy levels and disinterest in exercise (especially in a previously active individual) can be a symptom of depression. Social anxiety disorder, agarophobia, sleep disorders or substance abuse can all affect physical activity levels. Body image issues and self‐efficacy can likewise pose important psychological barriers that may require specific professional counselling and intervention to promote a more active lifestyle.
Medications
Although published research on this issue is limited, it is reasonable to assume that medications, which reduce energy levels, promote drowsiness, impair coordination or limit cardiorespiratory function can pose significant barriers to physical activity.
Now that we have discussed why it is important to asses the many factors that can affect energy metabolism, ingestive behaviour, and physical activity, in coming posts, we can explore how to apply this framework to patients presenting with weight gain.
@DrSharma
Edmonton, AB
Etiological Assessment of Obesity: Factors That Affect Physical Activity
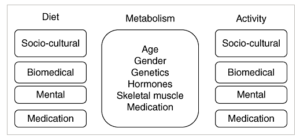 Continuing with citations from my article in Obesity Reviews on an aeteological framework for assessing obesity, we now turn to the some of the factors that can affect physical activity. Once we have established that weight gain in a given individual is not primarily driven by a change (decrease) in metabolic requirements, or primarily driven by ingestive behaviour, we turn to the issue of a decrease in physical activity as a drier of weight gain:
Continuing with citations from my article in Obesity Reviews on an aeteological framework for assessing obesity, we now turn to the some of the factors that can affect physical activity. Once we have established that weight gain in a given individual is not primarily driven by a change (decrease) in metabolic requirements, or primarily driven by ingestive behaviour, we turn to the issue of a decrease in physical activity as a drier of weight gain:
Barriers to Physical Activity
As with caloric intake, activity‐related caloric expenditure can vary from virtually zero (as in a bedridden individual) to several thousand calories a day (as in a competitive athlete). In considering physical activity, it is important to note that in sedentary individuals, the majority of activity thermogenesis results from non‐exercise activity thermogenesis (NEAT) simply from performing the acts of daily living, walking, posture and fidgeting. Any reduction in NEAT, even with no change in planned exercise frequency, duration or intensity, would result in reduced energy requirements. Evidence suggests that some individual’s resistance to weight gain is linked largely to their innate ability to spontaneously increase NEAT to defend against caloric excess.
As with nutrition, the factors that determine physical activity can be divided into four domains: socio‐cultural factors, biomedical factors, psychological factors and medications. Determining which of these domains is predominantly responsible for reduced physical activity or sedentariness can allow the clinician to specifically address those barriers in the management plan.
We will consider each of these factors in subsequent posts.
@DrSharma
Edmonton, AB


