Weight Effects of Beta-Blockade
Friday, February 11, 2011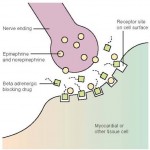 Almost exactly to this date 10 years ago, we published a paper in HYPERTENSION discussing the potential for weight gain associated with the use of beta (β) blockers (drugs that, at the time, were widely use for treating high blood pressure and presently continue to be routinely used for the management of coronary artery disease and heart failure).
Almost exactly to this date 10 years ago, we published a paper in HYPERTENSION discussing the potential for weight gain associated with the use of beta (β) blockers (drugs that, at the time, were widely use for treating high blood pressure and presently continue to be routinely used for the management of coronary artery disease and heart failure).
As we discussed in our paper, sympathetic activation is an important metabolic adaptation limiting weight gain and, therefore chronic β-blocker therapy could increase the propensity for weight gain by reducing energy expenditure.
This question was now readdressed by Lee and colleagues from Sydney, Australia, in a paper just published in the International Journal of Obesity.
In the current study, the reseachers first performed a mechanistic study comparing energy expenditure, diet-induced thermogenesis, and habitual activity levels between volunteers (n=11) with uncomplicated hypertension treated with a β-blocker and anthropometrically matched controls (n=19) and found that β-blocker treatment reduced diet-induced thermogenesis by around 50%, fat oxidation rate by 32% and weekly habitual activity levels by 30%.
They then examined data from three cross-sectional studies consisting of around 200 patients with diabetes, 80 patients with hypertension and over 11,000 participants in a large multi-centre diabetes trial (ADVANCE).
In all three populations, β-blocker treated patients had higher body weights: about 9 kg higher in the diabetes patients, around 17 kg higher in the hypertension patients, and around 5 kg higher in the ADVANCE trial.
The authors conclude that total energy expenditure (both from reduction in post-prandial thermogenesis and reduced activity levels is reduced and therefore body weight may significantly increase under chronic β-blocker use.
These findings must be interpreted with some caution, as ß-blockers consist of a very heterogeneous group of compounds with varying propensity for weight gain (e.g. carvedilol and nebivolol may have far less impact on weight than propranolol or atenolol).
Furthermore, this side-effect of ß-blockade should not distract from the fact that this class of drugs has saved 100s of 1000s of lives in patients with coronary artery disease or heart failure and should definitely not simply be discontinued in patients who need them.
On the other hand, clinicians should be aware of this side effect and advise patients to carefully monitor their weight to decrease the risk for weight gain – as always, prevention of weight gain is easier than treatment.
Fortunately, we now have a variety of other drugs for the treatment of uncomplicated hypertension that are not only weight neutral but are also devoid of other unwanted metabolic effects of ß-blockade.
AMS
Freising, Germany


