VBLOC Device Shows Promise for Diabetes Treatment
Wednesday, June 23, 2010Regular readers will recall previous posts on the VBLOC device, which uses intermittent electrical signals to block the vagal nerve thereby leading to weight loss. The previous post followed Enteromedic’s announcement that the early results from the EMPOWER study failed to quite meet its primary and secondary efficacy endpoints (resulting in a 70% fall in share price).
Yesterday, however, the share price of Enteromedics more than doubled after the company announced the follow-up results of two studies, to be presented in the coming days at the American Society for Metabolic and Bariatric Surgery Meeting in Las Vegas.
After soldiering on in the randomized, double-blind, controlled pivotal EMPOWER Study, participants now at 20 months experienced an almost 20% excess weight loss, while averaging 9 hours of device use per day.
Perhaps more interestingly, in another ongoing study (VBLOC-DM2 ENABLE), obese patients with Type-2 diabetes mellitus showed an improvement in HbA1c levels of 0.8% as well as a 25% excess weight loss over 12 months while averaging about 14 hours per day of therapy with the implantable Maestro RC System.
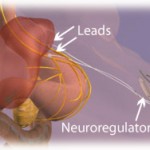
The novelty of this second generation Maestro device lies in the rechargeability of the implanted batteries via an external mobile charger and transmitter coil that can be worn for a few hours each week, thereby allowing delivery of a far more extensive treatment that with the previous version of the device.
Although this treatment required laparoscopic implantation of electrodes and the battery pack, with the usual risks entailed in such surgery, the treatment appears to be well tolerated with few side effects.
As outlined in yesterday’s press release, the company certainly appears optimistic about the future of this treatment for obesity and diabetes – the investors clearly appear to share this enthusiasm.
AMS
Edmonton, Alberta
p.s. You can now also follow me and post your comments on Facebook