Medical Barriers: Chronic Pain Conditions
 Today’s post is another excerpt from “Best Weight: A Practical Guide to Office-Based Weight Management“, recently published by the Canadian Obesity Network.
Today’s post is another excerpt from “Best Weight: A Practical Guide to Office-Based Weight Management“, recently published by the Canadian Obesity Network.
This guide is meant for health professionals dealing with obese clients and is NOT a self-management tool or weight-loss program. However, I assume that even general readers may find some of this material of interest.
CHRONIC PAIN CONDITIONS
Any condition that leads to chronic pain can contribute to obesity by increasing physiological and psychological stress. Pain also makes exercise more difficult to undertake and enjoy.
Osteoarthritis and Back Pain
Obesity is commonly associated with musculoskeletal pain and osteoarthritis, resulting in functional and motor limitations. Obese patients usually move more slowly, are less flexible and feel pain when performing tasks at floor level. There is a strong association between knee osteoarthritis and obesity. Degenerative arthritis resulting from chronic trauma associated with excess body weight develops primarily in weight-bearing joints such as the knee and ankle. However osteoarthritis can also be seen in non-weight-bearing joints, suggesting a systemic inflammatory response. Excess body weight is also closely related to lower back pain. Both static and compressive loading may damage the integrity of intervertebral discs. Increased biomechanical force can also cause muscle sprain and ligament strain.Sleep disorders are very prevalent among obese people. Obstructive sleep apnea is the most common disorder, but disturbed sleep may also be due to primary insomnia, or insomnia secondary to medications, medical or psychiatric disorders.
The presence of significant pain can promote immobility, leading to loss of muscle mass and reduced cardiopulmonary fitness. This can precipitate psychological and metabolic changes that promote further weight gain.
Patients with painful joints may benefit from water-based (non-weight-bearing) exercise and may require pain management before embarking on a weight-loss program. Obese patients requiring joint-replacement surgery are generally advised to lose weight (often in unrealistic amounts) before surgery, but this is very difficult if they are already partially immobilized by pain.
To complicate matters further, most commercial gyms are ill-equipped to handle the needs of obese clients, many of whom cannot go from lying down on the floor to standing up without assistance. Step classes and aerobics classes are in many cases ill-advised, and exercise machines and weight benches are not usually designed for obese users.
Fibromyalgia
Obesity is often associated with fibromyalgia, a common disorder characterized by fatigue, pain, stiffness in the trunk and extremities and a number of specific tender points. Fibromyalgia is more common in women than in men and is frequently accompanied by sleep disorders. Treatment of fibromyalgia may increase a patient’s ability to be physically active, and exercise has been shown to reduce the severity of fibromyalgia symptoms over time. As with severe osteoarthritis, starting with joint-sparing exercises may be prudent.
© Copyright 2010 by Dr. Arya M. Sharma and Dr. Yoni Freedhoff. All rights reserved.
The opinions in this book are those of the authors and do not represent those of the Canadian Obesity Network.
Members of the Canadian Obesity Network can download Best Weight for free.
Best Weight is also available at Amazon and Barnes & Nobles (part of the proceeds from all sales go to support the Canadian Obesity Network)
If you have already read Best Weight, please take a few minutes to leave a review on the Amazon or Barnes & Nobles website.
Why is it so Hard to Maintain a Reduced Body Weight?
 Yesterday, I had the pleasure of attending a lecture by Rudy Leibel from Columbia University, who is perhaps best known for his considerable contributions to our current understanding of energy metabolism.
Yesterday, I had the pleasure of attending a lecture by Rudy Leibel from Columbia University, who is perhaps best known for his considerable contributions to our current understanding of energy metabolism.
The talk was hosted by William Colmers as part of the University of Alberta Merck Translational Lecture Series.
In his presentation, Leibel addressed the issue of why it is so hard to keep weight off – in fact, even in people who undergo bariatric surgery, weight always comes back when surgery is reversed.
One of the key underlying problems is that when people lose weight, their energy expenditure does not simply fall to that of the energy expenditure of a person ‘naturally’ at that lower weight – it drops to levels far greater than expected.
Thus, a formerly-obese person burns 20% less calories than a never-obese person of that lower weight – or in other words a 200 lb person, who loses 40 lbs burns about 20% fewer calories than someone who is 160 lbs, but has never been obese. On top of this, the formerly-obese person experiences hunger, cold intolerance, and other behavioural and metabolic changes that make sustaining this lower body weight difficult.
From an evolutionary sense, this makes a lot of sense, as maintaining or ‘defending’ fat stores in the past has always been vital for human survival and therefore complex biological systems have evolved to readily take up and store excess calories when available and reduce caloric expenditure when times are tough.
In a large series of carefully conducted energy balance studies in humans, Leibel examined the impact of weight loss on energy expenditure, energy intake, neuroendocrine function, autonomic physiology, metabolism and brain imaging.
Whereas a short-term increase in body weight by 10 % results in a transient increase in energy expenditure, this returns to baseline, when the weight is lost. This means that weight-loss per se does not reduce energy expenditure.
On the other hand, a 10% drop in body weight immediately reduces energy expenditure by as much as 20%.
Interestingly, this fall in energy expenditure is not simply due to a fall in metabolic rate, but largely due to a decline in activity expenditure. This means that the body ‘saves’ energy not simply by turning down the furnace, but by becoming substantially more ‘fuel efficient’ during activity. In other words, someone who loses weight, will burn substantially fewer calories for a given amount of exercise than for the same amount of exercise performed before weight loss.
Much of this increase in ‘muscle efficiency’ can be attributed to the remarkable fall in the fat tissue-derived hormone leptin that occurs with weight loss.
Obese individuals apparently need a higher level of leptin to sustain energy balance. When they lose weight, thereby lowering their leptin levels, the system will aim to increase body fat levels to once again produce enough leptin to suppress the orexogenic response. Thus, weight reduction looks like ‘leptin deficiency’ to the brain, which it seeks to correct, by promoting weight gain.
The importance of leptin in this ‘defense’ response is clearly evident from both animal and human studies, in which leptin levels were maintained at or restored to pre-weight loss levels despite weight loss, by injecting leptin at levels just high enough to ‘mimic’ baseline levels.
In all of these experiments, using exogenous leptin to restore leptin levels to baseline, abolished the ‘defense’ mechanisms including the decline in total non-resting energy expenditure, thus making it easier to keep the weight off.
This ‘relative leptin-deficiency’ dependent improvement in muscle efficiency with weight loss can be clearly and consistently demostrated at the functional (exercise), imaging (MRI) and biological (biopsy) level.
Apart from reversing the improvements in muscle efficiency, Leibel also showed how the increased activation of hunger and appetite centres in the brain with weight loss can be reversed by leptin replacement. Thus, administration of leptin to individuals post-weight loss, reverses the decreased/delayed satiation and decreased perception of caloric density that would act to restore body weight to baseline.
Increases in the weight set-point occur with chronic weight gain, neuronal loss of aging, puberty and pregnancy. Unfortunately, lowering this threshold is far less likely, requiring such drastic measures as hypothalamic lesions or cachexia.
Thus, from an energy regulatory perspective, weight loss induces a ‘non-physiological’ state that can be restored to a ‘physiological’ state by leptin administration at levels high enough to mimic baseline levels.
So why is leptin not being sold to maintain weight loss? Because there is currently no regulatory pathway to license drugs that prevent weight regain. Regulators like the FDA and the EMEA simply lack a sound understanding of the complex physiology of weight regulation because after all, in weight management, the problem is never how to lose weight – the problem is always how to keep it off.
Unfortunately, based on the current guidelines for obesity drugs, there is no way for a pharma company to even apply to have a drug licensed that does not help reduce body weight (which leptin does not) but merely helps people keep weight off (which leptin does).
This is a shame, because in the end replacing leptin may well be the safest way to restore the ‘physiological’ state of being obese by correcting the ‘unphysiological’ state of having lost weight, which essentially drives weight regain.
AMS
Edmonton, Alberta
Obesity and Fibromyalgia
 Readers may recall previous posts on the relationship between obesity and fibromyalgia, a chronic condition characterised by widespread pain, muscle tenderness, and decreased pain threshold to pressure and other stimuli.
Readers may recall previous posts on the relationship between obesity and fibromyalgia, a chronic condition characterised by widespread pain, muscle tenderness, and decreased pain threshold to pressure and other stimuli.
Ursini and colleagues from Cantazaro, Italy, review the possible relationship between these two entities in a paper just published in Rheumatology International.
The authors review the epidemiological data showing that fibromyalgia patients have higher prevalence of obesity (40%) and overweight (30%) and discuss several mechanisms that may explain ‘the hidden link’ between these two conditions.
Proposed mechanisms include:
- impaired physical activity
- cognitive and sleep disturbances
- psychiatric comorbidity and depression
- thyroid dysfunction
- dysfunction of the GH/IGF-1 axis
- impairment of the endogenous opioid system
However, as they also note:
“…at this time is not possible to ascertain whether obesity is a cause or consequence of fibromyalgia.”
Nevertheless, the authors do suggest that the ‘causal’ relationship between the two conditions is supported by observations that fibromyalgia severity can be improved by weight loss.
This may be a good opportunity to remind readers that ‘causality’ is actually not always easy to demonstrate and it does take many levels of evidence to make any assumptions about causality convincing.
These include:
- Biological plausibility (this is usually where basic science comes in)
- Time course (this requires longitudinal studies)
- Dose-effect relationship (can be seen in cross-sectional studies)
- Strength of association (epidemiology)
- Coherence (consistency across different studies)
Add to this positive list the negative criterium that there must also be no ‘reasonable’ alternative explanations for the finding, and one may be able to eventually make a case for ‘causality’.
Unfortunately, medicine is fraught with uncertainity, as data from different studies may be conflicting or inconclusive.
This does not mean science is wrong – it just means that answers become more or less likely depending on the amount and quality of data and our understanding of the underlying biology.
This is the difference between science and pseudoscience – when new data emerges, scientists change their opinion – pseudoscientists ignore it.
Unfortunately, many prefer the ‘certainty’ of ignorance than the ‘uncertainty’ of true knowlegde.
This is why the progress of the ‘pilgrim of science’ is such an exciting and ever-fascinating journey.
I, for one, am glad that so many of my readers are willing to bear with me and join me on this expedition.
AMS
Edmonton, Alberta
Ursini F, Naty S, & Grembiale RD (2011). Fibromyalgia and obesity: the hidden link. Rheumatology international PMID: 21476098
Differential Gene Response to High-Fat Diets in Obesity
 Yesterday, as part of the kickoff of Live Right Now, CBC News published an extensive interview with me on why obesity is not simply a matter of “lifestyle choice”. As I point out in the the interview, “There’s a huge variability in how people can cope with extra calories…”
Yesterday, as part of the kickoff of Live Right Now, CBC News published an extensive interview with me on why obesity is not simply a matter of “lifestyle choice”. As I point out in the the interview, “There’s a huge variability in how people can cope with extra calories…”
Indeed, we have long known that people differ in their natural abilities to deal with caloric excess (or restriction) and that in some people metabolic and behavioural adaptations to changes in caloric intake can compensate for well over 500 calories a day with almost no change in body weight.
But there are not only differences in how the bodies deal with caloric excess – as blogged before, there are also important differences between individuals in the processing of and adaptations to changes in macronutrient composition of the diet.
This is, once again, demonstrated in a study by Kristen Boyle and colleagues from East Carolina University, just published online in the Journal of Clinical Endocrinology and Metabolism.
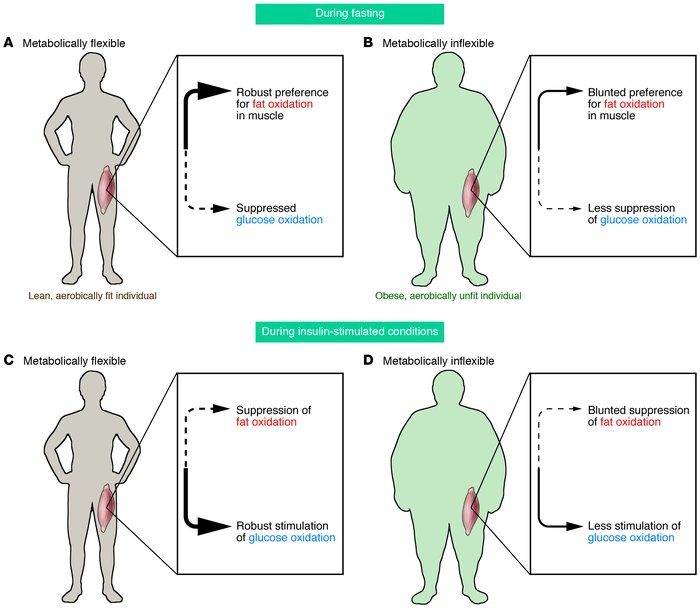
Based on the previous finding that lean individuals, in contrast to individuals with obesity, can increase whole body fat oxidation in response to a high-fat diet, the researchers examined the effect of increasing dietary fat intake on genes that regulate fat oxidation in skeletal muscle in both lean and obese insulin-resistant individuals.
A total of 12 lean (mean BMI = 22) and 10 obese (mean BMI = 40) individuals were studied before and after five days of a standardised high-fat diet.
While in lean individuals, skeletal muscle biopsies showed clear increases in the expression of genes for several key mediators of fat oxidation (e.g. pyruvate dehydrogenase kinase 4, uncoupling protein 3, PPARα, and PPARγ coactivator-1α), the expression of these genes remained unchanged or decreased in the obese participants.
Thus, this study shows a differential response to a lipid stimulus in the skeletal muscle of lean and insulin resistant obese humans.
Of course, this short-term study does not reveal whether or not these different metabolic responses to a high-fat diet are a cause or consequence of excess weight nor does this study provide any information on whether or not these metabolic patterns can be modified (e.g. through exercise).
Nevertheless, the study does support the notion that there is considerable variability in how people respond to changes in diet composition.
Perhaps these variations in metabolic response explain some of the controversy that I often hear from my patients (and as comments on this blog) – while some people swear that reducing carbs was the key to their success, others are convinced that their weight loss resulted largely from reducing their fat intake.
As I have said before, no single dietary strategy works for everyone and finding the right approach that works best for you or your patient may require some experimentation.
In weight management one size simply does not fit all!
AMS
Berlin, Germany
Boyle KE, Canham JP, Consitt LA, Zheng D, Koves TR, Gavin TP, Holbert D, Neufer PD, Ilkayeva O, Muoio DM, & Houmard JA (2010). A High-Fat Diet Elicits Differential Responses in Genes Coordinating Oxidative Metabolism in Skeletal Muscle of Lean and Obese Individuals. The Journal of clinical endocrinology and metabolism PMID: 21190973
Obesity And Multiple Sclerosis
 As blogged before, any medical condition that affects mobility, can potentially promote weight gain and the development of obesity with all its mental, physical and socioeconomic sequelae.
As blogged before, any medical condition that affects mobility, can potentially promote weight gain and the development of obesity with all its mental, physical and socioeconomic sequelae.
This of course also applies to patients diagnosed with multiple sclerosis (MS), a progressively disabling disease of the central nervous system currently affecting an estimated 2·5 million people worldwide. Multiple sclerosis is also the most common non-traumatic cause of disability in young adults.
In addition, patients with multiple sclerosis also often have psychiatric comorbidities ranging from depression, anxiety and social phobias to sleep disorders and chronic pain, which can all further adversely affect ingestive behaviour.
In a paper by Ruth Ann Marrie and Ralph Horowitz from the University of Manitoba, Winnipeg, Canada, just published in Lancet Neurology, the authors discuss why recognising obesity and other chronic illnesses in patients with multiples sclerosis is so important.
Apart from promoting the obvious sequelae of hypertension, diabetes and heart disease, obesity is also associated with delayed diagnosis of multiple sclerosis and obese patients tend to have higher levels of immobility and disability at the time of diagnosis.
Conversely, the diagnosis of comorbidities could be delayed by mistakenly attributing neurological symptoms such as progressive inability to walk, pain, or seizures to the pre-existing diagnosis of MS.
Corticosteroids used for relapses, may also contribute to weight gain and worsening of diabetes.
As the authors point out in the discussion:
“Further work is needed to establish whether pre-existing comorbidities affect the risk and phenotype of multiples sclerosis and, if so, how. Data from some studies indicate that comorbidities and health behaviours also affect disease progression. These findings need to be verified, and a broader range of disability outcomes, such as upper extremity and cognitive function, need to be taken into account. From a therapeutic perspective, important questions include whether comorbidities affect treatment choice, response, tolerability, and adherence, and whether we should use different treatment strategies in the presence of a comorbidity.”
As in all patients with impaired mobility and other physical or mental comorbidities, weight management can be challenging and should probably start well before weight gain sets in.
AMS
Edmonton, Alberta
To follow me and post your comments on Facebook click here
Marrie RA, & Horwitz RI (2010). Emerging effects of comorbidities on multiple sclerosis. Lancet neurology, 9 (8), 820-8 PMID: 20650403


