Proposed Functional Scoring in the Edmonton Obesity Staging System
 As regular readers may be well aware, the Edmonton Obesity Staging System (EOSS), now validated against several large prospective datasets including NHANES III, NHANES (1999–2004), and the Cooper Clinic Longitudinal Study, is a far better indicator of mortality risk in overweight and obese individuals than BMI, waist circumference, or even metabolic syndrome.
As regular readers may be well aware, the Edmonton Obesity Staging System (EOSS), now validated against several large prospective datasets including NHANES III, NHANES (1999–2004), and the Cooper Clinic Longitudinal Study, is a far better indicator of mortality risk in overweight and obese individuals than BMI, waist circumference, or even metabolic syndrome.
While determining EOSS stages for medical conditions such as hypertension, diabetes, or dyslipidemia is relatively straightforward and can be based on (more or less) accepted definitions within these conditions, scoring of physical function may be more complicated.
In our original proposal, we had recommended that functional limitations could be scored by the provider using clinical judgement as being either absent (Stage 0), mild (Stage 1), moderate (Stage 2), severe (Stage 3), or disabling/endstage (Stage 4).
However, I understand that for research (and perhaps even in clinical settings), one may wish to simplify and be more specific about what exactly entails ‘mild’, ‘moderate’, or ‘severe’.
In this context, I was quite intrigued by the findings of the impact of weight management on mobility in individuals with type 2 diabetes in the LookAHEAD study recently published in the New England Journal of Medicine.
Here is how the authors describe the assessment of mobility in that paper:
“Mobility was assessed on the basis of 6 of 11 items on the Medical Outcomes Study 36-Item Short-Form Health Survey (SF-36) Physical Functioning. The items included vigorous activity, such as running and lifting heavy objects; moderate activity, such as pushing a vacuum cleaner or playing golf; climbing one flight of stairs; bending, kneeling, or stooping; walking more than a mile; and walking one block. Participants were assigned a score of 1 on items for which they reported not being limited at all or a score of 0 on items for which they indicated having any limitation.”
Based on this assessment, the authors determined four states of disability with the following criteria:
“To render the model more clinically useful, it was reduced to four states that were sequential and progressively ordered from the healthiest to the most severe state of disability. In state 1 (good mobility), participants were somewhat unable to perform vigorous physical activities. In state 2 (mild mobility-related disability), participants had problems in bending and long-distance walking. In state 3 (moderate mobility-related disability), participants had deficits in many tasks and some deterioration in the ability to climb stairs and engage in moderately demanding activities. In state 4, participants had severe limitations, with difficulty in nearly all tasks.”
They further went on to describe the clinical relevance of these disability states:
“Using baseline data, we examined the clinical relevance of the four-state model. Moving from state 1 to state 4, the average body-mass index (BMI, the weight in kilograms divided by the square of the height in meters) increased progressively (33.83, 36.07, 37.39, and 38.79, respectively), as did the number of coexisting medical conditions (1.18, 1.44, 1.70, and 1.84). The estimated maximal MET capacity from state 1 to state 4 decreased linearly (8.16, 7.13, 6.52, and 5.94, respectively), and the ratio of women was disproportionately higher in state 4 than in state 1: although women constituted 50.0% of the good-mobility category, they constituted 72.0% of the severe-disability category.”
I believe, based on these findings, that these very criteria could be easily adopted for defining functional status in EOSS as follows:
EOSS Stage 0: no functional limitations
EOSS Stage 1: (good mobility) somewhat unable to perform vigorous physical activity (e.g. running and lifting heavy objects)
EOSS Stage 2: (mild mobility related disability) problems in bending and long-distance walking (>1 mile/1.5 Km)
EOSS Stage 3: (moderate mobility related disability) problems in climbing one set of stairs.
EOSS Stage 4: (severe mobility related disability) problems in walking one block.
How well these functional criteria overlap with other existing medical or mental comorbidities in overweight individuals and whether or not these functional criteria add to the discriminatory power of EOSS to determine mortality risk in overweight and obese patients, will need to be further validated and I would certainly be most interested in hearing from any readers who may perhaps wish to conduct such a research project or further validate this idea.
Clearly, functional limitations in overweight and obese individuals are not only common but can also impact other dimensions of health including physical fitness, mental health, and quality of life, and should therefore be taken into account in determining weight management strategies.
AMS
Edmonton, Alberta
Diet, Exercise, or Both for Knee Pain?
 The musculoskeletal consequences of overweight and obesity are perhaps by far the most disabling and costly sequelae of excess weight. Although they may be less likely to kill you than heart disease or stroke (also consequences of obesity), obesity related pains in your muscle, bones and joints can reduce your quality of life to a level where having a heart attack may seem like taking the easy way out.
The musculoskeletal consequences of overweight and obesity are perhaps by far the most disabling and costly sequelae of excess weight. Although they may be less likely to kill you than heart disease or stroke (also consequences of obesity), obesity related pains in your muscle, bones and joints can reduce your quality of life to a level where having a heart attack may seem like taking the easy way out.
So what is the best conservative treatment for weight-related knee pain?
This question was addressed now in a randomised controlled trial published last month in the British Medical Journal. Specifically, Claire Jenkinson and colleagues from the University of Nottingham, UK examined the effects of dietary intervention and quadriceps strengthening exercises on pain and function in overweight people with knee pain.
The study was performed in five general practices in Nottingham and involved 389 men and women aged 45 and over with a body mass index equal or greater than 28.0 and self reported knee pain.
Participants were randomised to dietary intervention plus quadriceps strengthening exercises; dietary intervention alone; quadriceps strengthening exercises alone; or and advice leaflet only (control group).
Dietary intervention consisted of individualised healthy eating advice that would reduce normal intake by 600 kcal a day. Interventions were delivered at home visits over a two year period.
In the 289 (74%) participants who completed the trial, patients in the knee exercise groups reported greater reduction in knee pain than in the non-exercise groups at 24 months, although the effect size was moderate (number needed to treat for a 30% improvement in knee pain was 9).
As expected the dietary intervention group lost some weight (around 3 Kg), but this degree of weight loss did not have any meaningful effect on knee pain or function but was associated with a reduction in depression.
The authors conclude that a home based, self managed programme of simple knee strengthening exercises over a two year period can significantly reduce knee pain and improve knee function in overweight and obese people with knee pain.
Although a moderate sustained weight loss is achievable with dietary intervention and is associated with reduced depression, it is not enough to influence pain or function.
As knee pain is a common problem in patients presenting with excess weight, which if not addressed, will ultimately result in immobility and increasing weight gain, exercise prescriptions should be routinely implemented in patients presenting with this problem.
AMS
Edmonton, Alberta
Spinal Fusion Surgery in Severe Obesity
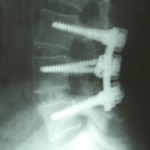 Lower back pain is not an uncommon finding in obese and very obese individuals. One surgical treatment option is to create a fusion between two or more vertebrae in an attempt to reduce pain by stopping the motion at the painful vertebral segment(s).
Lower back pain is not an uncommon finding in obese and very obese individuals. One surgical treatment option is to create a fusion between two or more vertebrae in an attempt to reduce pain by stopping the motion at the painful vertebral segment(s).
In a paper just published in SPINE, Rahul Vaidya and colleagues from the Detroit Receiving Hospital and University Health Center, Detroit, report on their experience in a case series of 63 patients with a BMI of 30 or higher.
Despite a higher surgical risk and a 45% greater chance of complications, obese and very obese patients showed significant improvement in visual analog scale for back and leg pain with some improvement in disability scores independent of the BMI of the patient.
Thus, despite posing a greater challenge for the surgeon and slightly higher surgical risk, heavier patients stand to benefit as much from surgery as less obese patients.
Incidentally, as with other types of orthopedic surgeries that improve mobility, no “spontaneous” weight loss was found to occur after spinal surgery.
Important questions that remain to be answered include the role for pre-surgical weight loss and whether or not weight management will be made easier following surgery.
AMS
Edmonton, Alberta
Pain in Older Obese Adults
BEST HEALTH BLOG FINALIST: The second round of voting is on – please vote AGAIN for your favourite health blog by clicking here
Last night I gave a talk to General Practitioners at the Medical School in Victoria, BC (hosted by Dr. Brad Amson, General/Bariatric Surgeon).
I spoke about the importance of carefully assessing patients for contributors and barriers to weight management.
As blogged yesterday, pain is one of the most common issues in patients with excess weight and can be both a contributor and barrier to weight gain and severely obese patients appear at particular risk for pain catastrophizing.
But how common is pain and how great is the incident risk in overweight and obese patients?
This issue was addressed in a recent study by Noor Heim and colleagues from the VU University, Amsterdam, The Netherlands, published in OBESITY.
This prospective study investigated the relationship between measured BMI and waist circumference with prevalent and incident pain in 2000 participants of the Longitudinal Aging Study Amsterdam, aged 55-85 years at baseline (1992-1993) and after after 3 years (N = 1,478) and 6 years (N = 1,271) of follow-up.
The overall prevalence of pain was 33% at baseline and increased significantly with higher quartiles of BM. After adjustment for age, education, depression, smoking, physical activity, and chronic diseases, both men and women in the highest quartile of BMI were around twice as likely to present with pain than individuals in the lowest BMI quartile.
Of the participants without pain at baseline, those in the highest quartile of BMI had a 2-fold increased odds for incident pain after 3 years of follow-up and around 2.5-fold after 6 years.
The association with BMI was independent of waist circumference, suggesting that absolute weight rather than weight distribution was important for pain.
This longitudinal study also answers the question whether or not pain in obesity is the chicken or the egg – it appears that obesity increases the risk for incident pain, rather than vice-versa.
But what about weight loss and its effect on pain? This issue, interestingly, has not been widely studied and results have been inconsistent. Apart from the rather dramatic improvement in pain and mobility reported in surgical obesity treatment, the effect of non-pharmacological or pharmacological weight loss on pain incidence or progression is not clear (though my guess is that weight loss can’t hurt – no pun intended).
Given the tendency of obese patients to catastrophize and the high incidence of depression and immobility, pain prevention and management is a key issue in the management of patients challenged by excess weight.
AMS
Victoria, BC
Pain Catastrophizing in Severe Obesity
BEST HEALTH BLOG FINALIST: The second round of voting is on – please vote AGAIN for your favourite health blog by clicking here
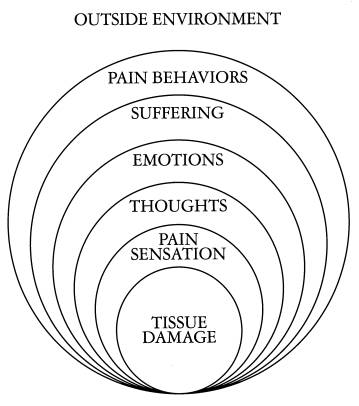 Pain is one of the most common and debilitating problems in patients challenged by severe obesity. Not just a consequence of mechanical complications of obesity (osteoarthritis, back pain, plantar fasciitis, fibromyalgia, etc.), pain is often a key barrier to physical activity and thus weight management. In fact, excess pain can promote psychological (e.g. depression, anxiety) and behavioural (e.g. binge eating) factors that may further promote weight gain.
Pain is one of the most common and debilitating problems in patients challenged by severe obesity. Not just a consequence of mechanical complications of obesity (osteoarthritis, back pain, plantar fasciitis, fibromyalgia, etc.), pain is often a key barrier to physical activity and thus weight management. In fact, excess pain can promote psychological (e.g. depression, anxiety) and behavioural (e.g. binge eating) factors that may further promote weight gain.
This issue is of even more importance in patients who display the now well-described phenomenon of pain catastrophizing, or the maladaptive responses to pain (tendency to focus on and magnify pain sensations with an intense sense of unbearable suffering and helplessness) that plays an extremely important role in how pain is perceived and processed. Pain catastrophizing now accounts for a substantial proportion of pain-related disability.
Studies in patients with fibromyalgia show that pain catastrophizing is associated with increased activity in brain areas related to anticipation of pain (medial frontal cortex, cerebellum), attention to pain (dorsal ACC, dorsolateral prefrontal cortex), emotional aspects of pain (claustrum, closely connected to amygdala) and motor control. Thus, catastrophizing influences pain perception through altering attention and anticipation, and heightening emotional responses to pain.
In another recent study by Tamara Somers and colleagues from Duke University, morbidly obese patients with osteoarthritis (OA) reported higher levels of pain catastrophizing than OA patients in the overweight and obese category. The severely obese patients who engaged in a high level of pain catastrophizing reported having much more intense and unpleasant pain, higher levels of binge eating, lower self-efficacy for controlling their eating and lower weight-related quality of life.
The relationship between pain catastrophizing and eating behaviour is of particular interest, as high-fat and high-sucrose foods have been shown to increase pain tolerance. Thus, binging on highly-palatable foods may be a compensatory response to emotional distress and pain. It is not difficult to see how patients can enter into a vicious cycle of pain, increased eating, weight gain, more pain, more eating, and so on.
In routine practice, pain catastrophizing can be easily and reliably assessed with questionnaires like this one, which can be scored like this.
Fortunately, pain catastrophizing is responsive to cognitive behavioural therapy, with clinically relevant improvements in upto 50% of individuals.
I would have little doubt that failure to recognize and address pain catastrophizing increases the likelihood of further weight gain and disability in patients with severe obesity.
AMS
Edmonton, Alberta



