Efficacy and Safety of Long-Term Low-Calorie Diet in Severely Obese Patients Non-Eligible for Surgery
 Low calorie diets (generally offering fewer than 1000 cal per day) have been been extensively used for the induction of medically supervised weight loss for decades. However, as with all diets, patients regain weight as soon as the diet is discontinued.
Low calorie diets (generally offering fewer than 1000 cal per day) have been been extensively used for the induction of medically supervised weight loss for decades. However, as with all diets, patients regain weight as soon as the diet is discontinued.
This raises the question of how long patients can safely stay on such a diet to sustain the often dramatic amounts of weight loss.
In a paper just published in Clinical Obesity, we describe the long-term efficacy and safety of using a low-calorie diet (LCD: providing 900 kcal per day) in severely obese patients who were non-eligible for bariatric surgery.
We describe eight patients (6 men, mean age 60) with severe obesity (mean BMI 57.1) who underwent long-term LCD (33 ± 10 months).
Variables of interest included anthropometric, cardiovascular risk and nutritional parameters, thyroid, renal and liver function, changes in medications, side effects and adverse events.
On average, patients lost 44 ± 15 kg (27 ± 13% of initial weight) at 24 months.
This dramatic weight loss (comparable to what can be achieved with bariatric surgery) resulted in substantial and sustained improvements in glucose homeostasis, blood pressure and lipid profile as well as dramatic improvements in mobility and quality of life.
Overall LCD was well tolerated with minor self-limited side effect (e.g constipation, dry skin, etc.). No nutritional deficiencies were noted.
Over the follow-up period, two subjects underwent coronary revascularization and one patient underwent knee replacement surgery – all recovered without complications.
These findings suggest that in selected obese patients, long-term management with LCD may provide an alternative option for substantial and sustainable weight loss with significant improvements in metabolic and cardiovascular health.
While most people may find it difficult (if not impossible) to give up all food to sustain themselves on a liquid LCD (4 shakes a day), this unique observation at least documents that for those rare individuals for whom there may be no other alternative, this can be a relatively safe and effective strategy for sustaining a substantial weight loss in the long-term.
@DrSharma
Kananaskis, AB
Liraglutide Helps Sustain Weight Loss
 While most people focus on the next big thing that will help them lose weight, the real unmet therapeutic need is for helping people keep the weight off.
While most people focus on the next big thing that will help them lose weight, the real unmet therapeutic need is for helping people keep the weight off.
This is why, I was particularly interested in a study by Tom Wadden and colleagues, just published in the International Journal of Obesity, which looked at the effect of treating individuals who had already lost weight with a low calorie diet with the GLP-1 analogue liraglutide (The SCALE™ trial).
The study was conducted in 422 obese participants, who managed to first lose at least 5% of their initial weight during a low-calorie (liquid) run-in diet (LCD), after which they were randomised to either 3.0 mg of liraglutide (once daily injections) or placebo.
During the 56 week trial, the liraglutide group lost an additionl 6% of their body weight (on top of the 6% they lost on the LCD), whereas the control group essentially maintained most of their initial weight loss.
Overall, liraglutide treated participants were about five times more likely to maintain at least 5% weight loss than those on placebo and about four times more likely to lose an addition 5% from their weight at time of randomisation.
As expected, the liraglutide treated group reported the typical gastrointestinal symptoms associated with GLP-1 treatment, although most of these events were apparently transient and mild or moderate in severity.
Thus, treatment with liraglutide (3.0 mg/day, together with diet and exercise), not only helped most of the participants maintain the weight loss achieved by LCD, but even further significantly reduce their weight over the 56 weeks of the trial.
Unfortunately, as noted in an earlier post, there is currently no regulatory pathway to license a drug for “weight loss maintenance”, which is why liraglutide will need to go the usual route and file for an indication as a “weight-loss” drug.
That said, it appears that once this drug is launched, it may well find a much-needed place in helping sustain and augment what can be achieved by a low-calorie diet.
@DrSharma
Edmonton, AB
Disclaimer: I have received speaking and consulting honoraria from Novo Nordisk, the maker of liraglutide. Liraglutide is currently not indicated or licensed for the treatment of obesity.
![]() Wadden T, Hollander P, Klein S, Niswender K, Woo V, Hale PM, & Aronne L (2013). Weight maintenance and additional weight loss with liraglutide after low-calorie diet-induced weight loss: the SCALE™ Maintenance randomized study. International journal of obesity (2005) PMID: 23812094
Wadden T, Hollander P, Klein S, Niswender K, Woo V, Hale PM, & Aronne L (2013). Weight maintenance and additional weight loss with liraglutide after low-calorie diet-induced weight loss: the SCALE™ Maintenance randomized study. International journal of obesity (2005) PMID: 23812094
.
Early Glycemic Benefits of Bariatric Surgery Are Largely Due to Caloric Restriction
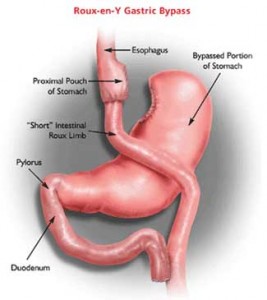 Currently, the notion that type 2 diabetes is somehow specifically “cured” by bariatric surgery (strategically renamed “metabolic surgery” by some) is receiving a lot of traction.
Currently, the notion that type 2 diabetes is somehow specifically “cured” by bariatric surgery (strategically renamed “metabolic surgery” by some) is receiving a lot of traction.
This is largely based on the observation that in many patients with diabetes, glycemic control rapidly improves within the first days of surgery (often even before significant weight loss sets in). Proponents of this “specific” benefit of surgery generally invoke the profound changes in gut hormones following bariatric surgery for these effects.
However, most bariatric clinicians will have observed similar “instantaneous” improvements in glycemic control in diabetic patients, who simply go on a fast or otherwise drastically restrict their caloric intake.
Now, a study by Mirjam Lips and colleagues from the Leiden University Medical Center in The Netherlands, published in Clinical Endocrinology suggests that most (if not all) of the (early) beneficial effect of bariatric surgery on glycemic control may be explained by the caloric restriction induced by this procedure.
In their study, Lips and colleagues, matched diabetic patients undergoing Roux-en-Y gastric bypass surgery with individuals undergoing the same degree of weight loss on a 600 KCal liquid very-low-calorie diet (VLCD). Both groups were exposed to a standardised meal challenge three weeks post-sugery or into the VLCD.
In both groups, baseline glycemic parameters as well as post-prandial glucose responses were similarly improved (essentially normalised in both groups).
Thus, caloric restriction was as effective in restoring early glycemic control in diabetic patients as was Roux-en-Y gastric surgery, suggesting that most of this effect was related to caloric restriction (and some weight loss) rather than any intrinsic mechanisms particular to surgery.
On the other hand, there were clear differences in the impact of surgery on incretins including increased levels of GLP-1 and PYY responses as well as greater suppression of ghrelin.
Based on these findings it appears that while the early beneficial effect of bariatric surgery on glycemic control may well be largely mediated by caloric restriction, the changes in gut hormones may well explain why patients with surgery are more successful in the long term in their ability to continue adhering to lower caloric intake and keeping the weight off.
AMS
Edmonton, AB
![]() Lips MA, de Groot GH, van Klinken J, Aarts E, Berends FJ, Janssen IM, Van Ramshorst B, Van Wagensveld BA, Swank DJ, Van Dielen F, van Dijk KW, & Pijl H (2013). Calorie restriction is a major determinant of the short-term metabolic effects of gastric bypass surgery in obese type 2 diabetic patients. Clinical endocrinology PMID: 23711328
Lips MA, de Groot GH, van Klinken J, Aarts E, Berends FJ, Janssen IM, Van Ramshorst B, Van Wagensveld BA, Swank DJ, Van Dielen F, van Dijk KW, & Pijl H (2013). Calorie restriction is a major determinant of the short-term metabolic effects of gastric bypass surgery in obese type 2 diabetic patients. Clinical endocrinology PMID: 23711328
.
Where Do Medically Supervised Low-Calorie Diets Fit In?
 Low calorie diets, usually consisting of a variable period of formula diets, followed by variable periods and intensities of follow-up, have been around for decades.
Low calorie diets, usually consisting of a variable period of formula diets, followed by variable periods and intensities of follow-up, have been around for decades.
While there is little doubt that such diets can help patients lose large amounts of weight in a short period of time (rivalling or even exceeding what is seen with bariatric surgery), the large-scale feasibility and durability of this intervention remains controversial.
A study that speaks both to the feasibility and efficacy, at least over a 1-3 year period, is now published by Stephan Bischoff (University of Hohenheim, Stuttgart, Germany) and colleagues in the latest issue of the International Journal of Obesity.
In this paper, Bischoff and colleagues describe the outcomes of a prospective longitudinal observational study of Prospective multicenter observational study in over 8,000 obese individuals (mean BMI 43.0) undergoing a medically supervised 52-week treatment in 37 centres across Germany (a small subset of patients was followed for up to 3 years).
The program consisted of 5 treatment phases:
Phase 1: a 1-weekintroduction time to check inclusion and exclusion criteria;
Phase 2: a 12-week-period of low-calorie diet (LCD; 800 kcal per day) during which participants consume formula diet exclusively (daily consumption of five packets of 160 kcal meal replacement products dissolved in 300 ml water each; Optifast 800 formula, Nestle´ Inc.), accompanied by 12 medical examinations, 12 exercise units, two behavior therapy lessons and two nutrition counselling sessions;
Phase 3: a 6-week-refeeding phase, during which solid food is reintroduced and formula diet is stepwise replaced by normal diet without change of total energy intake, accompanied by six medical examinations, six exercise units, two behaviour therapy lessons and six nutrition counselling session;
Phase 4: a 7-week stabilization phase in which energy intake is stepwise, enhanced to an individual level that allows weight stabilization, accompanied by three medical examinations, four exercise units, four behavior therapy lessons and three nutrition counselling session;
Phase 5: a 26-week-maintenance phase in which nutritional education and behavior modification is intensified to learn coping strategies and to achieve longterm weight control, accompanied by six medical examinations, 13 exercise units, 22 behavior therapy lessons and five nutrition counselling session.
Of the over 8,000 patients, who started the program, over 3,000 (or about 42%) discontinued the treatment.
Given this rather high discontinuation rate, the stated reasons for discontinuation are of interest: personal reasons (6.9%), no further appearance (6.5%), job related reasons (5.1%), disease/medical reasons (3.6%), financial reasons (3.4%), feeling of sufficient success (2.2%), familiar reasons (2.0%), mental/psychological reasons (1.5%), exclusion by the program team (1.4%), weight regain (1.0%), product dissatisfaction (0.6%) or pregnancy (0.4%). In about 1/6 of cases, the reason was unknown (7.2%).
Nevertheless, even with this relatively large number of discontinuations, the ‘intention-to-treat’ (ITT) results were impressive: At week 52, average weight loss was around 15 Kg in women and about 19 Kg in men. The prevalence of metabolic syndrome decreased by 50%. There were also remarkable improvements in quality of life and several other measures of health and functioning.
Overall the treatment was well tolerated with the most common ‘side effects’ being constipation, hair loss, and gall bladder problems (all known to be complication of significant weight loss).
So where do LCD interventions fit in?
The authors themselves are careful in pointing out that data on the long-term efficacy of such interventions are sketchy. Although there have been some reports that even several years after such an intervention, the average weight loss is still about 5% below the initial weight of the group, individuals weight trajectories appear highly variable. While some patients manage to keep much of the weight off, others will regain the weight within months of completing the intervention.
This should not come as a surprise given the chronicity of obesity – when treatment stops the weight comes back!
This then raises the issue of why one would even consider a time-limited obesity intervention program – why 52 weeks? Why not 104 weeks or 208 weeks or 416 weeks or in fact every week thereafter forever?
This is where things get murky. While I have no doubt that there are indeed a substantial number of individuals for whom an LCD approach works and may well lead to long-term weight loss maintenance (even with little long-term support or follow-up), I would assume that the vast majority of patients will require ongoing and lifelong continuing support – something seldom offered by such program and something that many patients are unlikely to comply with if too onerous or expensive.
This does not mean that there is not an important role for LCDs in bariatric practice. LCDs can certainly be considered a viable option for patients unwilling or unable to undergo bariatric surgery. They certainly also have their place in helping patients achieve rapid and medically safe weight loss in situations where such drastic weight loss is indicated (e.g. preparation for surgery or a diagnostic procedure).
Will such interventions prove to be cost effective? I believe the jury is out on this but this question clearly begs an answer.
As so often, the interpretation of the present findings will differ: while the ‘glass-half-empty’ folks will point to the high number of patients, who discontinue or rapidly regain the weight they lose, the ‘glass-half-full’ folks will highlight the fact that 2 in 3 patients of the 50% who do complete the program may still be well below their initial weight 3 years later.
Given our rather limited range of successful obesity treatment strategies and the fact that surgery may not be for everyone, I do strongly feel that LCDs should be part of the general armamentum of bariatric care and are probably underused in patients, who could well benefit from this approach and may well be overused in patients, who will probably not.
AMS
Edmonton, Alberta
![]() Bischoff SC, Damms-Machado A, Betz C, Herpertz S, Legenbauer T, Löw T, Wechsler JG, Bischoff G, Austel A, & Ellrott T (2011). Multicenter evaluation of an interdisciplinary 52-week weight loss program for obesity with regard to body weight, comorbidities and quality of life-a prospective study. International journal of obesity (2005) PMID: 21673653
Bischoff SC, Damms-Machado A, Betz C, Herpertz S, Legenbauer T, Löw T, Wechsler JG, Bischoff G, Austel A, & Ellrott T (2011). Multicenter evaluation of an interdisciplinary 52-week weight loss program for obesity with regard to body weight, comorbidities and quality of life-a prospective study. International journal of obesity (2005) PMID: 21673653
.
Nonsurgical Weight Loss for Extreme Obesity
 Yesterday’s post was about how we need to rethink and restructure obesity management in primary care. Today I discuss a primary care study that describes the outcome of non-surgical weight management in patients with extreme obesity.
Yesterday’s post was about how we need to rethink and restructure obesity management in primary care. Today I discuss a primary care study that describes the outcome of non-surgical weight management in patients with extreme obesity.
The paper by Donna Ryan and colleagues published in this week’s edition of the Archives of Internal Medicine describes the results of the Louisiana Obese Subjects Study (LOSS), a 2-year randomised, controlled, “pragmatic clinical trial” conducted in seven primary care practices and one research clinic.
Around 600 Volunteers with BMIs in the 40-60 range were screened and randomized to intensive medical intervention (IMI) (n = 200) or usual care (UCC) (n = 190). The IMI group recommendations included a 900-kcal liquid diet for 12 weeks or less, group behavioural counseling, structured diet, and choice of pharmacotherapy (sibutramine, orlistat, or diethylpropion) during months 3 to 7 and continued use of medications and maintenance strategies for months 8 to 24. In contrast, the UCC group received guidance in an internet weight management program.
The mean age of participants was 47 years; 83% were women, and 75% were white. Retention rates over two years were 51% for the IMI group and 46% for the UCC group. After 2 years, 31% in the IMI group achieved a 5% or more weight loss and 7% achieved a 20% weight loss or more, compared with 9% and 1% of those in the UCC group. A total of 101 IMI completers lost an average of –9.7% of their initial weight whereas weight in the 89 UCC completers remained virtually unchanged (which over 2 years is actually not such a bad result at all – remember, successful weight management starts with stopping the gain!).
While the study can no doubt be criticized for high attrition rates and relatively modest weight loss in IMI completers (only around 10% of initial weight), the study does show that at least for some patients, aggressive management strategies in primary care may provide sustainable outcomes that can have clear health benefits.
Let us not forget that attrition rates in disease management programs for other chronic diseases (e.g. diabetes, dysplipidemia, hypertension, etc.) are also relatively high and that only a minority of patients with these other common chronic conditions are ever fully controlled in primary care practice (despite the wide range of medical treatments and resources available to patients with these conditions).
Thus, there is no reason to believe that chronic disease management for obesity, when implement in primary practice, must necessarily fare worse than chronic disease management for other conditions. The fact that obesity management in primary practice appears so unsuccessful is not because interventions don’t work (this study shows they do), but rather because no serious attempt is made to address obesity in the first place.
While the 900-calorie liquid diet followed by intense behavioural and pharmacological treatment may not be everyone’s cup of tea, and of course comes nowhere near the results with bariatric surgery, for some patients this is may well be a safe and cost-effective strategy that can be delivered in primary practice.
Remember, in obesity treatment, one size certainly does not fit all and having a breadth of strategies rather than a single intervention is probably the only way to go.
I would certainly like to hear from anyone who has been on a 900-kcal liquid diet or who uses this approach in their patients.
AMS
Edmonton, Alberta


