Do Genetics Explain Variability in Weight-Loss Response to Liraglutide?
 As with any medication (for anything!) not everyone responds the same.
As with any medication (for anything!) not everyone responds the same.
Now a small study by Mojca Jensterle and colleagues from Ljubljana, published in the European Journal of Clinical Pharmacology, reports that genetic variability in the GLP-1 receptor gene may predict the variability to the human GLP-1 analogue liraglutide, now approved for obesity treatment in the US, Canada and Europe.
In their study, Jensterle and colleagues examine the realationship between two common alleles (variants) of the GLP-1 receptor in 57 women with obesity and polycystic ovary syndrome.
All women were treated with liraglutide 1.2 mg QD s.c (well under the 3.0 mg QD dose approved for obesity treatment) for 12 weeks.
Twenty of the participants were classified as strong responders (>5% weight loss), who lost about 7.4 Kg, whereas 37 were considered poor responders losing only 2.2 Kg.
Carriers of at least one rs10305420 allele were about 70% less likely to be a high responder than individuals with two wild-type alleles. Similarly, carriers of at least one rs6923761 allele were about three times as likely to high responders compared to homozygous carriers of the wild type.
Although my previous work in these type of genetic studies have made me highly critical (not to say sceptical) of these types of small studies, the notion that genetic variability in the GLP-1 receptor (the molecular target of liraglutide) may well lead to differences in response is not all that far fetched.
Thus, whether true or not, I have little doubt that indeed much of the variability in pharmacological response to liraglutide (or for that matter any other drug for anything) may well be determined by genetics.
Whether testing people for genetic markers before starting a specific treatment will ever become reality for obesity and whether or not, the genetic variability seen in this study will still be seen when lirglutide is used at the actual dose approved for obesity treatment remains to be seen.
In the meantime, the easiest way to see who responds and who does not is to try it. This why the regulatory approval of liraglutide for obesity comes with a simple stopping rule – if it doesn’t work for you – stop taking it!
@DrSharma
Edmonton, Alberta
Disclaimer: I have received consulting and speaking honoraria from Novo Nordisk, the maker of liraglutide.
How Your Gut Tastes What You Eat
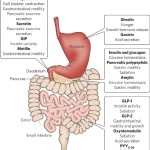 If you thought that the only senses that determine the palatability of food are your sense of taste and smell, you may be wrong.
If you thought that the only senses that determine the palatability of food are your sense of taste and smell, you may be wrong.
It turns out that we have a rather sophisticated sensing mechanism in our gut that senses the composition of our diet and interacts with the brain to regulate our appetite and food intake.
Just how exactly this gut “nutrient-sensing” system works, is reviewed by Sophie Hamr and colleagues from the University of Toronto in a paper published in Current Diabetes Reports.
As the authors point out,
“…the gastrointestinal (GI) tract is anatomically positioned to provide initial feedback following a meal via detection of incoming nutrients and relaying signals to the brain and peripheral tissues to prevent excess energy intake and circulating nutrients…..This, coupled with the vast neural and humoral connectivity of the gut to other important sites of energy regulation, such as the brain, allows the gut to effectively relay information to the rest of the body about the size and composition of an incoming meal.”
Each nutrient (fats, carbohydrates, protein) interacts with specific sensory and signal transduction mechanisms in the gut.
Animal studies show that exposing the gut to certain nutrients (for e.g. by tube feeding) can stimulate or suppress feeding behaviour making animals chose or avoid certain foods. Often these effects can persist for days or even weeks, well beyond the time course of a single meal.
Furthermore, these effects appear to be largely dependent on the presence of specific nutrients rather than on the actual nutritional or energy state of the animal.
“…these evidences lend notion for the intestine to sense specific nutrients (i.e., lipid and carbohydrate) at specific concentrations, rather than calories, in an effort to drive further food consumption.”
The authors point out that changes in how the gut senses nutrients may well explain how bariatric surgery works to reduce appetite and change food preferences.
No doubt, a better understanding these mechanisms and the molecular mechanisms involved could lead to novel dietary or pharmacological interventions to prevent or treat obesity.
@DrSharma
Edmonton, AB
Post-Surgery Weight Regain: Hormonal and Metabolic Factors
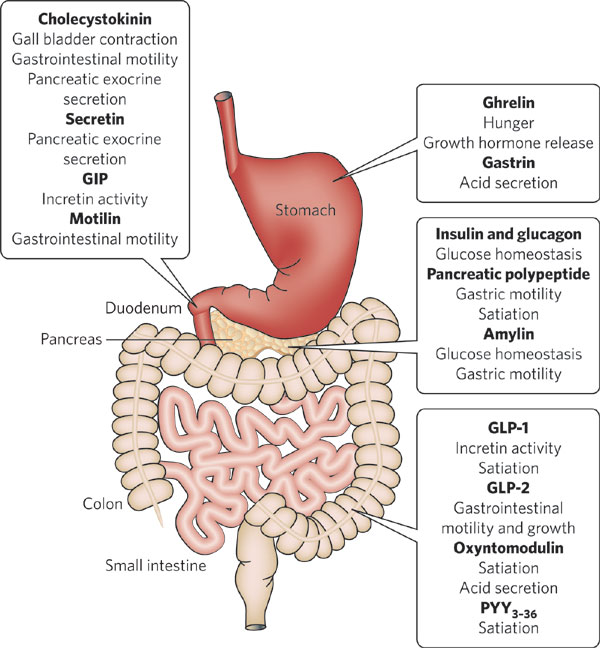 In yesterday’s post, I discussed the importance of dietary factors in weight regain after bariatric surgery.
In yesterday’s post, I discussed the importance of dietary factors in weight regain after bariatric surgery.
In this post, I will discuss the role of hormonal and metabolic factors identified in our systematic review of post-surgical weight regain published in Obesity Surgery.
It is now widely assumed that the efficacy of bariatric surgery is not solely dependent on causing a “restriction” or simply “malabsorbtion” of calories.
Rather, there is now growing consensus that the key reason why bariatric surgery works is through its impact on gut hormones and neurological signals from the gut that significantly reduce hunger and/or satiety.
Thus, it is not surprising that in our review we found several studies that noted a significant relationship between post-surgical levels of the “hunger hormone” ghrelin and post-surgical weight regain. Patients who experienced less weight loss or greater weight regain after sleeve gastrectomy and/or roux-en-y bypass surgery demonstrated higher fasting and post-prandial ghrelin levels. Elevated ghrelin levels were also found to correlate with a return of hunger in patients with regain.
Other evidence points to the role of hypoglycaemia in promoting weight regain in some patients. Reactive hypoglycaemia after bariatric surgery may result from the rapid transit of ingested carbohydrates into the small intestine thereby generating an early and significant insulin surge which results in a reactive hypoglycaemia shortly after a meal. This would in turn prompt snacking and increased caloric ingestion resulting in weight regain.
While there is currently no medical treatment to deal with ghrelin elevations, the latter problem can potentially be managed by dietary measures, including the avoidance of high-glycemic index foods.
Given that there are many gut hormones that may be altered by bariatric surgery and their individual roles are still poorly understood, it is clear that we will need further studies to better understand how these factors may explain why some patients failure to lose weight after surgery or show a greater tendency for weight regain.
@DrSharma
Chicago, IL
![]()
Karmali S, Brar B, Shi X, Sharma AM, de Gara C, & Birch DW (2013). Weight Recidivism Post-Bariatric Surgery: A Systematic Review. Obesity surgery PMID: 23996349
.
Does Bariatric Surgery Work By Changing Gut Bugs?
 As readers are well aware, bariatric surgery is currently the most effective treatment for severe obesity. Yet, we are still not quite sure how exactly surgery works.
As readers are well aware, bariatric surgery is currently the most effective treatment for severe obesity. Yet, we are still not quite sure how exactly surgery works.
For one, we know that simplistic notions of surgery “mechanically” inhibiting food intake are simply wrong – were that the case, simply having your jaws wired shut would be as effective.
Nor does caloric malabsorption account for much of the weight loss (except perhaps with biliopancreatic diversion) – there is indeed good evidence that with contemporary bypass procedures, the remaining gut has no problem extracting all the calories it needs from the diet.
One of the reasons bariatric surgery works in the long term is because it changes gut hormones (e.g. ghrelin, GLP-1, PYY) and thus counteracts the homeostatic mechanisms that counteract weight loss.
But recent observations that bariatric surgery also prevents much of the decrease in metabolic rate normally associated with weight loss suggests that there is more going on than meets the eye.
Now, a study by Ling-Chun Kong and colleagues from Paris, France, in a paper published in the American Journal of Clinical Nutrition supports the notion that bariatric surgery may have additional effects on energy homeostasis by affecting the gut microbiome.
The researchers profiled the gut microbiota from fecal samples and adipose tissue samples in severely obese individuals, before as well as three and six months after roux-en-Y gastric bypass surgery.
In these patients, surgery resulted in a remarkable increase in richness of gut microbiota (previous studies have shown that gut microbiota phylogenetic richness is lower in obese than in lean subjects), whereby almost 40% of the increase in gut bacteria belonged to the phylum Proteobacteria. Most of these changes occurred early, with no further differences noted between the three and six month samples.
The researchers also found significant associations between gut microbiota composition and adipose tissue gene expression (including numerous genes related to metabolism and inflammation) as well as clinical phenotype – as substantial proportion of which were independent of any changes in caloric intake.
As to the cause or clinical implications of these changes, the authors can only speculate. As they point out, some of these changes may be due to alterations in diet composition, eating behaviour (e.g. increased chewing), or biological changes that include differences in pH and other aspects of intestinal milieu.
Although such “correlational” studies cannot prove cause and effect, this study does document profound changes in the gut bacteriome with gastric bypass surgery and given our understanding that gut bugs may very much influence energy metabolism, it will be certainly be of considerable interest to examine whether these changes contribute to the success of bariatric surgery.
AMS
Edmonton, AB
![]() Kong LC, Tap J, Aron-Wisnewsky J, Pelloux V, Basdevant A, Bouillot JL, Zucker JD, Doré J, & Clément K (2013). Gut microbiota after gastric bypass in human obesity: increased richness and associations of bacterial genera with adipose tissue genes. The American journal of clinical nutrition PMID: 23719559
Kong LC, Tap J, Aron-Wisnewsky J, Pelloux V, Basdevant A, Bouillot JL, Zucker JD, Doré J, & Clément K (2013). Gut microbiota after gastric bypass in human obesity: increased richness and associations of bacterial genera with adipose tissue genes. The American journal of clinical nutrition PMID: 23719559
.
Early Glycemic Benefits of Bariatric Surgery Are Largely Due to Caloric Restriction
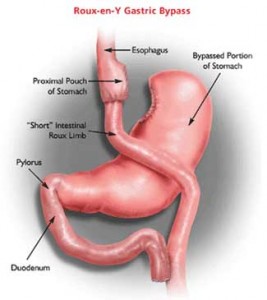 Currently, the notion that type 2 diabetes is somehow specifically “cured” by bariatric surgery (strategically renamed “metabolic surgery” by some) is receiving a lot of traction.
Currently, the notion that type 2 diabetes is somehow specifically “cured” by bariatric surgery (strategically renamed “metabolic surgery” by some) is receiving a lot of traction.
This is largely based on the observation that in many patients with diabetes, glycemic control rapidly improves within the first days of surgery (often even before significant weight loss sets in). Proponents of this “specific” benefit of surgery generally invoke the profound changes in gut hormones following bariatric surgery for these effects.
However, most bariatric clinicians will have observed similar “instantaneous” improvements in glycemic control in diabetic patients, who simply go on a fast or otherwise drastically restrict their caloric intake.
Now, a study by Mirjam Lips and colleagues from the Leiden University Medical Center in The Netherlands, published in Clinical Endocrinology suggests that most (if not all) of the (early) beneficial effect of bariatric surgery on glycemic control may be explained by the caloric restriction induced by this procedure.
In their study, Lips and colleagues, matched diabetic patients undergoing Roux-en-Y gastric bypass surgery with individuals undergoing the same degree of weight loss on a 600 KCal liquid very-low-calorie diet (VLCD). Both groups were exposed to a standardised meal challenge three weeks post-sugery or into the VLCD.
In both groups, baseline glycemic parameters as well as post-prandial glucose responses were similarly improved (essentially normalised in both groups).
Thus, caloric restriction was as effective in restoring early glycemic control in diabetic patients as was Roux-en-Y gastric surgery, suggesting that most of this effect was related to caloric restriction (and some weight loss) rather than any intrinsic mechanisms particular to surgery.
On the other hand, there were clear differences in the impact of surgery on incretins including increased levels of GLP-1 and PYY responses as well as greater suppression of ghrelin.
Based on these findings it appears that while the early beneficial effect of bariatric surgery on glycemic control may well be largely mediated by caloric restriction, the changes in gut hormones may well explain why patients with surgery are more successful in the long term in their ability to continue adhering to lower caloric intake and keeping the weight off.
AMS
Edmonton, AB
![]() Lips MA, de Groot GH, van Klinken J, Aarts E, Berends FJ, Janssen IM, Van Ramshorst B, Van Wagensveld BA, Swank DJ, Van Dielen F, van Dijk KW, & Pijl H (2013). Calorie restriction is a major determinant of the short-term metabolic effects of gastric bypass surgery in obese type 2 diabetic patients. Clinical endocrinology PMID: 23711328
Lips MA, de Groot GH, van Klinken J, Aarts E, Berends FJ, Janssen IM, Van Ramshorst B, Van Wagensveld BA, Swank DJ, Van Dielen F, van Dijk KW, & Pijl H (2013). Calorie restriction is a major determinant of the short-term metabolic effects of gastric bypass surgery in obese type 2 diabetic patients. Clinical endocrinology PMID: 23711328
.


