Will Immunotherapy Provide a New Treatment for Obesity?
 Obesity is widely associated with low grade inflammation and previous studies have noted increased incidence of allergic responses in obese individuals pointing to a possible role of immune response in the regulation of metabolism and body weight.
Obesity is widely associated with low grade inflammation and previous studies have noted increased incidence of allergic responses in obese individuals pointing to a possible role of immune response in the regulation of metabolism and body weight.
Last week Nature Medicine published three separate articles, which together may well herald in an entirely new area of obesity research: immunometabolism or metabolic immunology. Although all studies reports finding in mouse models of obesity and diabetes, their observations may well have important implications for human obesity.
In the first study, Satoshi Nishimura and colleagues from the University of Tokyo show that CD8+ effector T cells play an important role in macrophage recruitment and adipose tissue inflammation. They not only found that large numbers of CD8+ T cells infiltrated obese epididymal adipose tissue in mice fed a high-fat diet (while there was a depletion of CD4+ T ells), but also that this infiltration preceded the accumulation of macrophages in fat tissue. On the other hand immunological and genetic depletion of CD8+ T cells lowered macrophage infiltration and adipose tissue inflammation and ameliorated systemic insulin resistance. Coculture and other in vitro experiments revealed a vicious cycle of interactions between CD8+ T cells, macrophages and adipose tissue. These results support the notion that CD8+ T cells have an essential role in the initiation and propagation of adipose inflammation.
In the second study, Markus Feuerer and colleagues from Harvard Medical School, Boston, USA, showed that CD4+ regulatory T cells with a unique phenotype were highly enriched in the abdominal fat of normal mice, but, similar to the observation by Nishimura and colleagues, their numbers were strikingly and specifically reduced in insulin-resistant models of obesity. Loss-of-function and gain-of-function experiments revealed that these regulatory T cells influenced the inflammatory state of adipose tissue, insulin resistance, and glucose uptake.
In the third study, Shawn Winer and colleagues from The Hospital for Sick Children, University of Toronto, Canada, showed that immunotherapy with CD4+ (but not CD8+) T cell transfer into lymphocyte-free obese mice reversed weight gain and insulin resistance. In other obese mice, brief treatment with CD3-specific antibody or its F(ab’)2 fragment reversed insulin resistance for months, despite continuation of a high-fat diet.
Together these papers not only document the important role of immune response in the development of obesity and its complications but also identify a number of novel targets and strategies that could be harnessed to treat obesity in manners similar to the treatment of other immunological abnormalities including allergies.
The studies certainly lend credence to those who have long suggested that obesity may be related to allergic or other immunological responses to allergens and environmental toxins.
How these findings will translate into better treatments for obesity obviously remains to be seen.
AMS
Edmonton, Alberta
Are Obese Kids Prone to Allergies?
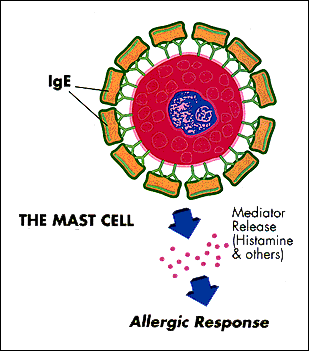 Several studies have suggested a link between the increasing rates of obesity and the increasing rates of allergies. However, much about this link remains doubtful.
Several studies have suggested a link between the increasing rates of obesity and the increasing rates of allergies. However, much about this link remains doubtful.
The largest study on this issue was recently published by Cynthia Visness and colleagues from the University of Chapel Hill, North Carolina, USA in the Journal of Allergy and Clinical Immunology.
In this study, Visness and colleagues examined data from the 2005-2006 National Health and Nutrition Examination Survey (NHANES), which included data from over 4000 kids aged 2-19. Allergies were determined both by a symptom questionnaire on previous diagnoses of hay fever, eczema, and allergies as well as measurement of allergen-specific serum IgE levels.
Obese kids had significantly (~30%) higher total IgE levels and a ~60% higher rate of food sensitization compared to normal-weight kids. C-reactive protein levels were also higher in obese kids suggesting signs of systemic inflammation. It is however important to note that there was no relationship between obesity and reported allergy symptoms or hay fever
Nevertheless, the authors conclude that there may indeed be a higher risk for (food-related) allergies in obese kids.
Obviously, there are limitations to the interpretation of these findings. As this is a cross-sectional study, no inference can be made on what causes what – does obesity contribute to food allergies (obese kids have been shown to eat a greater variety of foods – see yesterdays post in this regard) – or do food allergies contribute to obesity? Or are both obesity and allergies related to some common phenomenon such as reduced breast feeding, and simply associated without being causally linked?
Obviously, as we do not know if the relationship is causal, it would be premature to suggest that preventing or treating obesity would reduce allergies or that treating allergies would reduce the risk for obesity – this can only be shown by appropriately designed intervention studies.
AMS
Edmonton, Alberta


