Blood Glucose Levels Modulate Neural Control of Appetite
Tuesday, November 1, 2011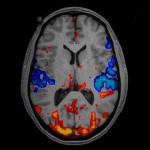 Anyone, who has ever experienced even a mild drop in blood glucose levels, understands the notion of hunger – a drive so powerful, that almost any food will taste good (energy-dense foods will taste even better!).
Anyone, who has ever experienced even a mild drop in blood glucose levels, understands the notion of hunger – a drive so powerful, that almost any food will taste good (energy-dense foods will taste even better!).
But whether or not elevated glucose levels can also suppress appetite is less well studied.
As study by Kathleen Page and colleagues from Yale University, just published in the Journal of Clinical Investigation, examines whether obese and nonobese individuals regulate their desire to consume high-calorie foods differently in response to changes in blood glucose levels.
Using functional MRI (fMRI) combined with a stepped hyperinsulinemic euglycemic-hypoglycemic clamp, that allows changing the blood glucose levels in a controlled fashion, the authors show that even modest reductions in blood glucose levels preferentially activate limbic-striatal brain regions in response to food cues to produce a greater desire for high-calorie foods.
In contrast, high-normal blood glucose levels preferentially activated the medial prefrontal cortex, an area of the brain involved in regulating impulse control and reducing motivation for rewarding stimuli such as food and drugs.
Interestingly, however, higher circulating glucose levels predicted greater medial prefrontal cortex activation only in lean but not in obese subjects.
As the authors discuss:
“These results are consistent with reports showing that high BMI is associated with decreased prefrontal activity at rest and after meal consumption and that obese subjects have an attenuated postprandial deactivation of the hypothalamus. These altered obesity-associated neural responses to food cues may contribute to overeating behavior, especially several hours after consumption of high-carbohydrate meals, a time when glucose often declines significantly below baseline levels.”
Thus, as the authors conclude:
“These findings demonstrate that circulating glucose modulates neural stimulatory and inhibitory control over food motivation and suggest that this glucose-linked restraining influence is lost in obesity.”
They also speculate that:
“Strategies that temper postprandial reductions in glucose levels might reduce the risk of overeating, particularly in environments inundated with visual cues of high-calorie foods.”
One strategy to avoid drops in blood glucose levels is not to allow yourself to go hungry by consuming smaller but more frequent meals. The other is perhaps to chose low-glycemic index foods in order to prevent the ‘crash-and-crave’ drive that follows rapid changes in blood glucose levels.
The study, certainly provides further evidence for important ‘biological’ differences between non-obese and obese people – while the former experience ‘natural’ appetite suppression with high-normal glucose levels, the
latter do not experience such a suppression of appetite and will need to resort to conscious restraint – a far more difficult undertaking.
AMS
Edmonton, Alberta
p.s. Hat tip to Bill Graber for pointing me to this study
Page KA, Seo D, Belfort-Deaguiar R, Lacadie C, Dzuira J, Naik S, Amarnath S, Constable RT, Sherwin RS, & Sinha R (2011). Circulating glucose levels modulate neural control of desire for high-calorie foods in humans. The Journal of clinical investigation, 121 (10), 4161-9 PMID: 21926468


