How The Body Weighs Itself – Evidence For A Bone “Gravitostat”
Wednesday, January 3, 2018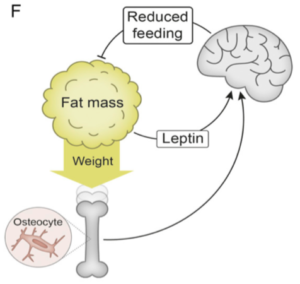 In my talks, I have often joked about how to best keep weight off – just carry around a backpack that contains the lost pounds to fool the body into thinking the weight is still there.
In my talks, I have often joked about how to best keep weight off – just carry around a backpack that contains the lost pounds to fool the body into thinking the weight is still there.
It turns out that what was intended as a joke, may in fact not be all too far from how the body actually regulates body weight.
As readers of these posts are well aware, body weight is tightly controlled by a complex neuroendocrine feedback system that effectively defends the body against weight loss (and somewhat, albeit less efficiently, protects against excessive weight gain).
Countless animal experiments (and human observations) show that following weight loss, more often than not, body weight is regained, generally precisely to the level of initial weight.
With the discovery of leptin in the early 90s, an important afferent part of this feedback system became clear. Loss of fat mass leads to a substantial decrease in leptin levels, which in turn results in increased appetite and decreased metabolic rate, both favouring weight regain and thus, restoration of body weight to initial levels.
Now, an international team of researchers led by John-Olov Jansson from the University of Gothenburg, Sweden, in a paper published in the Proceeding of the National Academy of Science (PNAS), provides compelling evidence for the existence of another afferent signal involved in body weight regulation – one derived from weight-bearing bones.
Prompted by observations that prolonged sedentariness can promote weight gain, independent of physical activity, the researchers hypothesised that,
“…there is a homeostat in the lower extremities regulating body weight with an impact on fat mass. Such a homeostat would (together with leptin) ensure sufficient whole body energy depots but still protect land-living animals from becoming too heavy. A prerequisite for such homeostatic regulation of body weight is that the integration center, which may be in the brain, receives afferent information from a body weight sensor. Thereafter, the integration center may adjust the body weight by acting on an effector.”
In a first series of experiments, the researchers observed that implanting a weight corresponding to about 15% of body weight into rodents (rats and mice), resulted in a rapid “spontaneous” adjustment in body weight so that the combined weight of the animal plus the weight implant corresponded more-or-less to that of control animals.
Within two weeks of implanting the weights, ∼80% of the increased loading was counteracted by reduced biological weight, largely due to reduced white adipose tissue (WAT), accompanied by a corresponding decrease in serum leptin levels. (Interestingly, this weight loss was also accompanied by a substantial improvement in insulin resistance and glucose homeostasis).
The decrease in “biological” body weight was mainly attributable to a reduction in caloric intake with no changes in fat oxidation, energy expenditure or physical activity.
Removal of the implanted weights resulted in rapid weight regain to initial levels, showing that the “weight sensor” was active in both directions.
Experiments showed that this “weight sensing” mechanism was largely independent of the leptin pathway and did not appear to involve grehlin, GLP-1, a-MSH, estrogen receptor-a, or the sympathetic nervous system.
Now for the interesting part: the observed effect of weight loading was not seen in mice depleted specifically of DMP1 osteocytes, demonstrating that the suppression of body weight by loading is dependent on osteocytes.
As the authors note, these findings are consistent with a growing body of data indicating that the skeleton is an endocrine organ that regulates energy and glucose metabolism. Indeed, it is well known that osteocytes can sense dynamic short term high-impact bone loading for local bone adaptation – now it appears, that osteocytes may also play a vital role in sensing overall body weight and signalling this to the brain centres that regulate energy balance and body weight.
Thus, in summary, not only have the authors provided compelling evidence for a “weight-sensing” role for bone osteocytes (presumably through their presence in the long weight-bearing bones of our lower extremities) but also provide a plausible biological explanation for the weight gain and change in fat mass seen with prolonged sedentariness (which literally takes the weight off the bone).
These findings may also finally explain why rats held at increased gravity for extended periods of time (simulated G2) become lean even when their energy intake matches their expenditure.
Perhaps, carrying around a heavy backpack may indeed help with long-term weight loss maintenance after all – who knew?
@DrSharma
Berlin, D
Hat tip to Jean-Philippe Chaput for alerting me to this article


